Welcome to your ultimate guide on aluminum valence electrons! If you've ever been puzzled by the atomic structure of aluminum and wondered how it behaves in chemical reactions, this guide is for you. Understanding valence electrons is crucial for anyone studying chemistry, whether you're a student or a professional in the field. Our aim is to break down complex concepts into simple, actionable steps, backed by real-world examples and practical solutions.
Problem-Solution Opening Addressing User Needs
Many of us grapple with the fundamental yet complex concepts in chemistry, particularly the idea of valence electrons. Aluminum, being the third most abundant element in the Earth's crust, plays a pivotal role in various applications from manufacturing to everyday household products. Knowing how to determine and understand the valence electrons of aluminum can significantly boost your comprehension of chemical bonding and reactivity. However, the dense scientific jargon often obscures the practical aspects that can make this topic more approachable and interesting. Our guide aims to simplify these intricate details, offering you clear, step-by-step instructions that demystify aluminum's valence electrons.
Quick Reference
Quick Reference
- Immediate action item: To determine aluminum's valence electrons, look at its position in the periodic table.
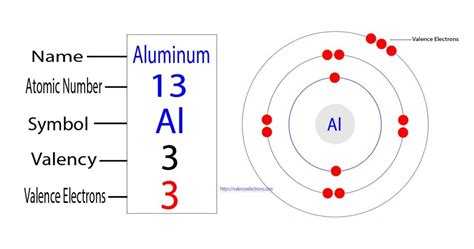
- Essential tip: Aluminum is in group 13, which means it has three valence electrons.
- Common mistake to avoid: Don't confuse valence electrons with total electrons. Remember, aluminum has 13 electrons in total, but we’re only interested in those in the outermost shell.
Detailed How-To Sections
Understanding Atomic Structure
To fully grasp aluminum’s valence electrons, we first need to understand atomic structure. Atoms consist of protons, neutrons, and electrons. Electrons occupy different energy levels around the nucleus, the innermost of which are the core electrons, and the outermost are the valence electrons.
An atom’s position in the periodic table reveals much about its electronic configuration. Aluminum (symbol Al) is found in group 13 and period 3. Its atomic number is 13, indicating it has 13 electrons distributed among these energy levels.
The electron configuration of aluminum is written as 1s2 2s2 2p6 3s2 3p1. Here, each set of numbers and letters represents an electron shell and its subshell. To focus on the valence electrons, we look at the outermost shell, which in aluminum’s case is the third shell (3s2 3p1).
Determining Valence Electrons
For any element, valence electrons are the electrons in the outermost shell. For aluminum, you see that the third shell (3s2 3p1) contains three electrons. These electrons are critical in forming chemical bonds and thus influence aluminum’s reactivity and its role in various chemical reactions.
Here’s a step-by-step guide:
- Identify the group: Aluminum belongs to group 13.
- Count the valence electrons: Group number equals the number of valence electrons. Therefore, aluminum has three valence electrons.
Practical Examples
Let’s put this knowledge into practice with real-world examples:
- Corrosion Resistance: Aluminum’s valence electrons are pivotal in its reaction with oxygen, forming a protective oxide layer. This process shields the bulk of the metal, preventing further oxidation and offering excellent corrosion resistance.
- Conductivity: The free movement of valence electrons in aluminum makes it an excellent conductor of electricity, which is why it’s used extensively in electrical wiring.
Practical FAQ
Why is it important to know the number of valence electrons in aluminum?
Knowing the number of valence electrons in aluminum helps in predicting its chemical behavior. For instance, knowing that aluminum has three valence electrons explains its tendency to lose these electrons to achieve a stable electron configuration, making it reactive with metals and non-metals in various industrial and everyday applications.
Advanced Insights
For those looking to delve deeper, understanding how aluminum’s valence electrons interact with other elements can lead to advanced applications in materials science. Here, the formation of aluminum compounds like aluminum chloride (AlCl3) showcases how it interacts with other elements to form stable compounds. Mastery of this concept can significantly aid in designing new materials with desired properties.
This guide has navigated you through the fundamental concepts to provide a comprehensive understanding of aluminum’s valence electrons. Whether for academic purposes or professional use, this detailed insight aims to equip you with the knowledge to explore the atomic world with confidence and clarity.