Understanding the intricacies of average atomic mass calculations is crucial for chemists and scientists alike. This aspect of chemistry plays a significant role in various scientific disciplines, including biochemistry, environmental science, and material science. This article delves into the fundamental principles and real-world applications, offering practical insights into this essential calculation.
The Fundamentals of Average Atomic Mass
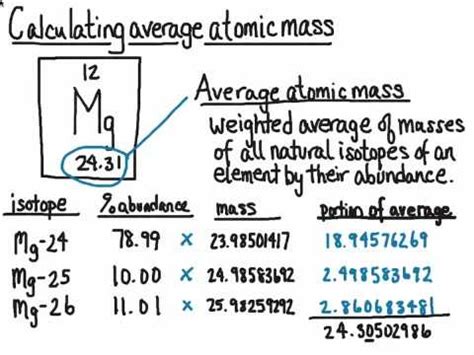
Average atomic mass, also known as mean atomic mass, is a weighted average of the masses of the atoms of an element, taking into account the relative abundance of each isotope. This concept is derived from the natural occurrence of different isotopes, which are variants of an element with varying numbers of neutrons.To illustrate, consider carbon, which predominantly consists of two stable isotopes: Carbon-12 and Carbon-13. Carbon-12 constitutes about 98.93% of natural carbon, while Carbon-13 makes up approximately 1.07%. The average atomic mass is calculated using the formula:
Average Atomic Mass = (Relative abundance of Isotope 1 x Mass of Isotope 1) + (Relative abundance of Isotope 2 x Mass of Isotope 2) +... + (Relative abundance of Isotope n x Mass of Isotope n)
Key Insights
- Primary insight with practical relevance: Accurate calculation of average atomic mass is crucial for determining molecular formulas and chemical reactions.
- Technical consideration with clear application: Understanding isotopic abundances enables precise quantification of compounds.
- Actionable recommendation: Always refer to the latest periodic table for up-to-date isotopic data.
Real-World Applications
The calculation of average atomic mass is not merely an academic exercise; it has profound implications in various scientific fields. For instance, in biochemistry, precise isotopic composition is pivotal for tracing metabolic pathways and understanding enzyme functions. Accurate atomic mass calculations enable researchers to determine the molecular weight of biomolecules, which is essential for drug design and development.Moreover, in environmental science, the isotopic composition of samples can reveal the origins of pollutants and trace atmospheric changes over time. In material science, the precise control of isotopic ratios in materials impacts the performance and longevity of devices, from semiconductors to nuclear reactors.
Calculation Techniques and Pitfalls
Calculating average atomic mass requires meticulous attention to detail. A common pitfall is misinterpreting the relative abundances of isotopes. Always ensure that the percentages are expressed in decimal form before inserting them into the formula. Another challenge arises in the availability of isotopic data; certain elements may have more recent data available from databases such as the National Institute of Standards and Technology (NIST).Consider the example of chlorine, which has two stable isotopes: Chlorine-35 and Chlorine-37. The relative abundance of Chlorine-35 is approximately 75.78%, while Chlorine-37 makes up about 24.22%. The average atomic mass is calculated as follows:
Average Atomic Mass of Chlorine = (0.7578 x 34.96885) + (0.2422 x 36.96590) = 35.4527
Can average atomic mass differ from the atomic weight listed on the periodic table?
Yes, the atomic weight on the periodic table is often an approximation based on the most recent and precise average atomic mass calculations from reliable scientific sources.
Why is precision in isotopic abundances essential?
Precision is crucial because small errors in isotopic abundances can lead to significant discrepancies in calculated average atomic masses, affecting molecular formulas and chemical reactions.
By mastering the calculations of average atomic mass, scientists and chemists can ensure more accurate and reliable results across their respective domains. The importance of this fundamental principle cannot be overstated in the pursuit of scientific excellence.