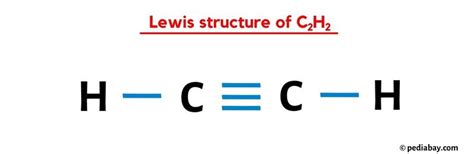
A comprehensive understanding of the C2H2 (acetylene) Lewis structure is fundamental for anyone delving into organic chemistry or molecular bonding theories. This molecule, also known as ethyne, provides a compelling study in the principles of covalent bonding, molecular geometry, and resonance structures.
The acetylene molecule consists of two carbon atoms triple-bonded to each other and each carbon atom single-bonded to a hydrogen atom. This configuration demands a careful examination of electron distribution and bonding. To grasp these intricacies, it’s essential to dive deep into the Lewis structure concept, which offers a visual representation of valence electrons and their participation in bonding.
Key Insights
- Primary insight with practical relevance: Understanding the C2H2 Lewis structure is crucial for elucidating its electronic characteristics, which affect its reactivity and interactions.
- Technical consideration with clear application: The linear geometry and triple bond length in acetylene illustrate how linear molecules can be formed in sp hybridized carbon atoms.
- Actionable recommendation: To effectively draw the C2H2 Lewis structure, start by correctly placing the least electronegative atom in the center and distribute the valence electrons accordingly.
Understanding Electron Distribution
The C2H2 molecule’s Lewis structure starts with identifying the total valence electrons. Each carbon atom has four valence electrons, and each hydrogen contributes one. Thus, C2H2 has a total of ten valence electrons. The carbon atoms should be bonded to each other with a triple bond, which consists of three pairs of electrons. Additionally, each carbon atom forms a single bond with a hydrogen atom, utilizing one electron pair per bond.
This arrangement optimizes electron distribution, ensuring that all atoms achieve a stable configuration, typically aiming for an octet (except hydrogen, which achieves a duet). The triple bond between the carbon atoms involves two pi bonds, contributing to the molecule’s overall stability and unique reactivity.
Geometry and Hybridization
The Lewis structure of C2H2 also leads us to understand its molecular geometry. The triple bond restricts the molecule’s geometry to be linear, with a bond angle of 180 degrees. This linearity is a result of the sp hybridization of the carbon atoms, which allows the formation of two sigma bonds and leaves two p-orbitals to form the pi bonds.
This sp hybridization creates an elongated structure with linear geometry, minimizing electron repulsion and resulting in a very stable configuration. This understanding not only explains acetylene’s molecular geometry but also parallels its physical and chemical properties.
Can acetylene exhibit resonance structures?
While acetylene (C2H2) does not exhibit resonance in the conventional sense due to the fixed triple bond, its stability can be partially attributed to electron delocalization within the triple bond. Resonance structures are generally observed in molecules with multiple double bonds that can shift positions, which is not the case with acetylene’s triple bond.
How does the C2H2 structure affect its chemical properties?
The triple bond in acetylene, represented by its C2H2 structure, contributes to high reactivity, making it a versatile compound in organic synthesis and industrial applications. It is also less saturated compared to alkanes and alkenes, which affects its stability and tendency to participate in addition reactions.
This definitive guide on the C2H2 Lewis structure offers a critical understanding of molecular bonding and geometry in organic chemistry. The linear structure, triple bond, and sp hybridization are foundational concepts that extend beyond acetylene, influencing a wide range of chemical behaviors and reactions in more complex molecules.