Understanding the geometry of molecules is critical in chemistry because it dictates how molecules interact with each other and influences their chemical properties. Many students and professionals struggle to grasp the different molecular shapes, but with structured guidance, it can become an intuitive aspect of chemistry. This guide is designed to help you master molecular shapes, breaking down complex concepts into actionable advice with real-world examples.
Mastering Molecular Shapes: Your Journey Starts Here
Imagine trying to bake a cake without a recipe or understanding the structure of a building without architectural plans. Both would be nearly impossible. In chemistry, molecular shapes provide the framework for understanding how atoms arrange themselves in molecules. The geometry of a molecule affects its properties, including reactivity, polarity, and physical characteristics. Understanding these shapes is not just an academic exercise; it has practical implications in fields ranging from pharmaceuticals to materials science.
Whether you're a student grappling with your first chemistry course or a professional aiming to deepen your expertise, this guide will help you navigate the world of molecular geometry with step-by-step instructions, practical solutions, and expert tips. Let’s embark on this journey to make molecular shapes a part of your chemistry toolkit.
Quick Reference: Get Started with Key Concepts
Quick Reference
- Immediate action item with clear benefit: Start with understanding the VSEPR theory (Valence Shell Electron Pair Repulsion) which is foundational to predicting molecular geometry.
- Essential tip with step-by-step guidance: To determine molecular shape, count the number of electron groups around the central atom and use the VSEPR model to predict geometry.
- Common mistake to avoid with solution: Misidentifying the number of lone pairs can lead to incorrect molecular shape. Always account for both bonding and non-bonding electrons around the central atom.
Understanding VSEPR Theory: The Foundation
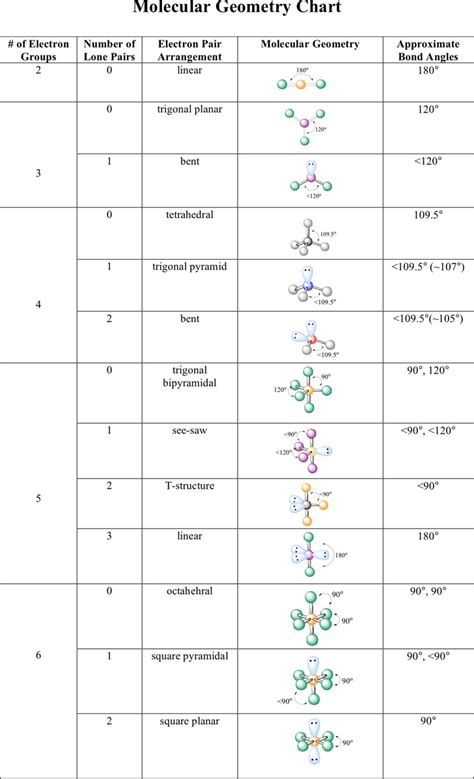
The VSEPR (Valence Shell Electron Pair Repulsion) theory is the cornerstone for predicting molecular shapes. According to this theory, electron pairs around a central atom repel each other and arrange themselves to minimize repulsion. Here’s a step-by-step approach to master this theory:
1. Determine the central atom: This is usually the atom with the lowest electronegativity unless specified otherwise. In molecules like CO₂, carbon is the central atom.
2. Count the valence shell electron pairs: For the molecule CF₄, carbon has four valence electrons, and since it bonds with four fluorine atoms, there are no lone pairs. Thus, there are four total electron groups.
3. Identify the electron group geometry: Based on the number of electron groups, identify the geometry. For four groups, the electron geometry is tetrahedral.
4. Convert to molecular geometry: Since there are no lone pairs, the molecular geometry is also tetrahedral. However, for more complex molecules, lone pairs may alter this geometry.
5. Apply to real molecules: Consider H₂O. Oxygen has six valence electrons, forms two bonds with hydrogen, and has two lone pairs. There are four electron groups, resulting in a tetrahedral electron geometry but bent molecular shape due to lone pairs.
Following these steps with practice will solidify your understanding of VSEPR theory and molecular geometry.
Detailed How-To: Predict Molecular Shapes with Ease
Predicting molecular shapes involves more than just the VSEPR theory; it requires detailed application and understanding of different molecular configurations. Here's a comprehensive how-to guide:
1. Learn the basic shapes: Understand the basic shapes: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
| Molecular Shape | Electron Groups | Geometry |
|---|---|---|
| Linear | 2 | 180° angle |
| Trigonal Planar | 3 | 120° angle |
| Tetrahedral | 4 | 109.5° angle |
| Trigonal Bipyramidal | 5 | Two different angles (90°, 120°) |
| Octahedral | 6 | 90° angle |
2. Identify the central atom: It’s typically the least electronegative atom or the one bonded to multiple atoms.
3. Determine electron groups: Electron groups include both bonding pairs and lone pairs. For NH₃, nitrogen is the central atom bonded to three hydrogens with one lone pair.
4. Use the VSEPR table: Match the number of electron groups to the VSEPR table to determine the electron geometry. For NH₃, there are four electron groups, indicating tetrahedral electron geometry.
5. Account for lone pairs: If there are lone pairs, they affect the molecular geometry. For NH₃, the lone pair makes the molecular shape trigonal pyramidal instead of tetrahedral.
6. Practice with examples: Use diverse molecules to practice. Consider SF₆, where sulfur forms six bonds with fluorine with no lone pairs, resulting in an octahedral electron and molecular geometry.
Practical FAQ: Your Most Asked Questions Answered
Why is molecular shape important?
Molecular shape is crucial because it directly affects the molecule’s properties such as polarity, reactivity, and how it interacts with other molecules. For instance, water’s bent shape contributes to its high polarity and unique properties like hydrogen bonding, influencing its role in biological systems.
What if a molecule has lone pairs?
Lone pairs occupy more space than bonding pairs due to higher repulsion, leading to distortions in the molecular shape. For example, in NH₃, the lone pair on nitrogen distorts the three N-H bonds, making the molecular geometry trigonal pyramidal rather than tetrahedral.
Can I use VSEPR theory for all molecules?
While VSEPR theory is very useful, it has its limitations. It works well for most molecules but might not perfectly describe molecules with extensive resonance structures or those that have significant ionic character. For complex molecules, additional considerations may be necessary.
By following this guide and practicing with different molecules, you’ll master the geometry of molecules, enabling you to predict their properties and behaviors accurately. Remember, practice and repetition are key to internalizing these concepts.
With this understanding, you’re now equipped to delve deeper into molecular chemistry and appreciate the intricate beauty of molecular shapes!