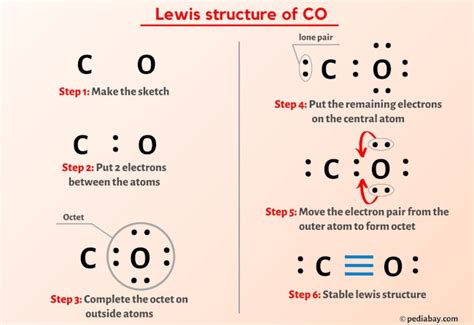
Understanding the CO (carbon monoxide) Lewis structure is crucial for grasping foundational concepts in chemistry, particularly when dealing with covalent bonding and molecular geometry. CO, a simple diatomic molecule, presents an intriguing case that can serve as a cornerstone for more complex structures. This guide will help demystify the Lewis structure for CO, ensuring you get the most out of it in practical chemistry applications.
Addressing the Need for CO Lewis Structure Mastery
If you’re a student or professional dealing with chemical bonding, the concept of the Lewis structure is fundamental. For CO, a molecule you’ll encounter in both academic and applied chemistry, mastering its Lewis structure is pivotal. Missteps here can lead to incorrect interpretations of chemical behavior, reactivity, and even molecular properties. This guide will walk you through the essential steps to draw an accurate CO Lewis structure, using practical examples to cement your understanding and avoid common pitfalls.
Quick Reference
Quick Reference
- Immediate action item: Count valence electrons. Carbon has 4, and Oxygen has 6.
- Essential tip: Always aim to satisfy the octet rule for each atom in your structure.
- Common mistake to avoid: Don’t forget that double or triple bonds are often necessary to achieve a full octet.
Drawing the CO Lewis Structure
To construct the CO Lewis structure, follow these steps:
- Step 1: Calculate total valence electrons. Carbon has 4 valence electrons and Oxygen has 6, making a total of 10 valence electrons for CO.
- Step 2: Place the least electronegative atom in the center. In CO, Carbon is less electronegative than Oxygen, so it will be the central atom.
- Step 3: Distribute electrons to form bonds. Start by placing two pairs of electrons between Carbon and Oxygen to form a double bond. This uses up 4 of the 10 valence electrons.
- Step 4: Complete the octets. Carbon now has 4 electrons around it (2 from the double bond), and Oxygen has 6 (4 from bonding plus 2 non-bonding pairs). Neither needs further electrons, as Carbon with only 4 electrons usually forms a double bond in CO, while Oxygen satisfies its octet with 8 electrons.
Your CO Lewis structure should depict a carbon atom double-bonded to an oxygen atom, with no lone pairs remaining.
Advanced Considerations in CO Lewis Structure
Once you’ve mastered the basics, you can explore more complex aspects of the CO molecule. Understanding formal charges and resonance structures can deepen your comprehension:
- Step 1: Calculate formal charges to verify your structure. For CO, the formal charge on Carbon is 0, and on Oxygen, it’s also 0. This indicates a balanced structure.
- Step 2: Consider resonance structures. While CO primarily displays one structure, understanding resonance can provide insight into its stability and reactivity.
- Step 3: Examine molecular geometry. Although CO is linear due to sp hybridization, visualizing this can help in understanding the molecule’s properties and behavior in reactions.
Practical FAQ
Why is it important to complete the octets correctly?
Completing the octets correctly is vital for predicting the molecule’s stability and reactivity. Following the octet rule ensures that each atom has a full valence shell, minimizing electron repulsion and maximizing bonding strength.
What if there are not enough electrons to complete the octet?
If there aren’t enough electrons to complete the octet for all atoms, consider using multiple bonds (double or triple) or pushing electrons to form a negative charge on more electronegative atoms. In CO, Carbon typically forms a double bond with Oxygen to achieve a full octet.
How does the CO Lewis structure relate to its reactivity?
The Lewis structure of CO helps predict its reactivity. With a double bond, CO is less likely to form further covalent bonds, making it less reactive than molecules with single bonds. However, it’s highly reactive with metals due to its ability to act as a ligand, binding to metal atoms in coordination complexes.
By following this guide, you’ll not only be able to accurately draw the CO Lewis structure but also understand its implications for molecular behavior and reactivity. This foundation will support more advanced studies in chemistry, providing a clear pathway from basic concepts to complex applications.