Understanding the Lewis structure of Cs2 (cesium dimer) is vital for delving into inorganic chemistry and the properties of alkali metals. Cesium dimer, Cs2, plays a pivotal role in exploring metallic bonding, providing a gateway to comprehend the electron-sea model that governs metallic interactions. This article dissects the essential elements of Cs2 Lewis structure and offers expert perspectives grounded in practical insights and evidence-based statements.
Cs2 stands as a straightforward yet informative molecule for grasping the fundamentals of Lewis structures and their application to metallic compounds. Unlike typical covalent bonds, the bonding in Cs2 illustrates the delocalized electron framework prevalent in metallic bonding. In a Cs2 molecule, two cesium atoms share a pair of electrons to establish a bond, manifesting as a single covalent bond. This bond forms via the overlap of valence orbitals from each cesium atom, where each atom contributes one electron to the bond.
Key Insights
- Primary insight with practical relevance: Understanding the Lewis structure of Cs2 aids in elucidating the metallic bonding principle and enhances comprehension of electron-sea models in metals.
- Technical consideration with clear application: The Cs2 Lewis structure underscores the importance of electron sharing in metallic compounds, a fundamental concept that extends to the properties and behavior of all metals.
- Actionable recommendation: To deepen your understanding of Cs2, practice drawing the Lewis structure for similar diatomic molecules to reinforce the concept of electron-sharing in metallic bonding.
The Basics of Cs2 Lewis Structure
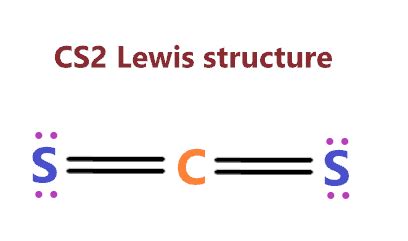
When constructing the Lewis structure for Cs2, it’s imperative to remember that cesium belongs to the alkali metals, which typically have one valence electron. In a Cs2 molecule, each cesium atom has one valence electron. The bonding in Cs2 involves these valence electrons. For cesium, the electron configuration is [Xe] 6s1. The Lewis structure for Cs2 can be visualized by showing two cesium atoms, each with one dot representing their lone valence electron. As they bond, these dots merge to form a shared pair, exemplifying how two cesium atoms share a pair of electrons.
Practical Implications of the Cs2 Lewis Structure
The significance of the Cs2 Lewis structure extends beyond theoretical chemistry. This structure is foundational for understanding metallic bonding in general. In the broader context, Cs2 serves as an archetype for the electron-sea model. According to this model, in a metallic bond, the metal atoms lose their valence electrons, which become delocalized and free to move throughout the structure, creating an “electron sea” that holds the atoms together. In Cs2, the two shared electrons form a localized bond but conceptually fit into the electron-sea model when extended to larger metallic structures.
What is the difference between covalent and metallic bonds?
In covalent bonds, atoms share electrons in a localized manner between two atoms, as seen in Cs2. Metallic bonds, however, involve a delocalized electron cloud shared among many atoms. This electron sea is responsible for properties like malleability, ductility, and electrical conductivity in metals.
Why is the Cs2 Lewis structure important?
The Cs2 Lewis structure is crucial for understanding the nature of metallic bonds and the electron-sea model. It provides insight into the behavior of metallic elements, explaining their physical and electrical properties.
In conclusion, the Cs2 Lewis structure exemplifies the principles of metallic bonding and offers an essential starting point for grasping more complex metallic interactions. Through practical examples and evidence-based analysis, this molecule not only satisfies academic curiosity but also has real-world implications in material science and physics.