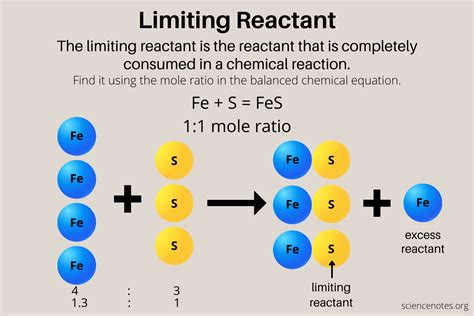
In the realm of chemical reactions, the concept of the limiting reactant plays a pivotal role in determining the outcome. Understanding this concept is essential for both theoretical and practical applications in chemistry. The limiting reactant is the substance that dictates the extent of a chemical reaction; once it is completely consumed, the reaction cannot proceed further regardless of the amount of other reactants present. This principle helps in calculating the maximum yield of products and managing resources effectively.
Defining the Limiting Reactant
A limiting reactant, also known as the limiting reagent, is the reactant that is entirely used up first in a chemical reaction. It effectively limits the amount of products that can be formed in the reaction. By identifying the limiting reactant, chemists can predict the amount of product that will form and ensure that no excess reagents go to waste. For instance, consider a reaction between hydrogen and oxygen to form water:
2H2 + O2 → 2H2O
If you mix a certain amount of hydrogen gas with an excess of oxygen gas, the hydrogen will be the limiting reactant if it runs out first during the reaction. This reaction would then produce twice the amount of hydrogen in water, based on the initial hydrogen provided.
Key Insights
- The limiting reactant determines the maximum amount of product that can be formed in a chemical reaction.
- When identifying the limiting reactant, calculate the moles of each reactant and compare their ratios with the stoichiometry of the balanced equation.
- Use the limiting reactant to calculate the theoretical yield of the product.
Importance in Stoichiometry
In stoichiometry, the limiting reactant is crucial for determining the amounts of reactants and products. When balancing a chemical equation, stoichiometric coefficients provide the mole ratio of reactants and products. If there’s an excess of one reactant, determining which one limits the product formation involves a few straightforward steps. First, convert all reactant amounts to moles, then use the coefficients from the balanced equation to find the theoretical yield of each product for each reactant. The reactant that yields the least amount of product when fully consumed is the limiting reactant.
Applications in Industrial Chemistry
Beyond laboratory experiments, identifying the limiting reactant has significant industrial implications. In manufacturing processes, precise control over reactants is essential to maximize output and minimize waste. For example, in the production of fertilizers, the stoichiometric ratio of nitrogen to phosphorus dictates the yield of the final product. If nitrogen is supplied in excess but phosphorus runs out first, phosphorus becomes the limiting reactant, and it governs the amount of fertilizer produced.
FAQ Section
How can I identify the limiting reactant in a chemical equation?
To identify the limiting reactant, convert all reactants to moles. Then, compare the mole ratios of each reactant to the stoichiometric coefficients in the balanced chemical equation. The reactant that yields the least product is the limiting reactant.
Can there be more than one limiting reactant?
In most reactions, there is typically a single limiting reactant. However, in complex multi-step reactions or if there are multiple reactants in varying stoichiometries, a thorough analysis is needed to determine which reactant limits the overall reaction progress.
Understanding the limiting reactant is fundamental for accurate product yield predictions, efficient resource management, and optimized chemical processes. Whether in the lab or on an industrial scale, this concept ensures that chemists and engineers can work towards the most efficient and cost-effective outcomes in their respective fields.