Understanding the differences between galvanic and electrolytic cells is crucial for professionals working in fields like chemistry, electrical engineering, and materials science. This article delves into the core differences and practical applications of these two types of cells. Whether you’re a student, a researcher, or an industry professional, this guide provides valuable insights to help you make informed decisions based on evidence-based statements and real-world examples.
Key Insights
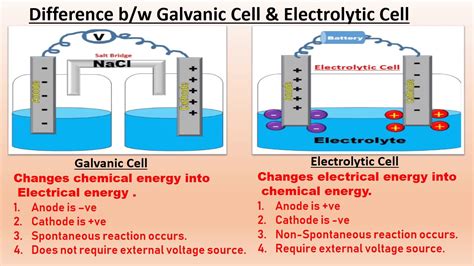
- Galvanic cells convert chemical energy into electrical energy through spontaneous redox reactions.
- Electrolytic cells use electrical energy to drive non-spontaneous redox reactions, essential for various industrial processes.
- For sustainable applications, opt for galvanic cells in scenarios where energy efficiency is paramount.
The Fundamentals of Galvanic Cells
Galvanic cells, also known as voltaic cells, are designed to convert chemical energy into electrical energy through spontaneous redox reactions. The core principle revolves around the flow of electrons from a site of lower chemical potential to a site of higher chemical potential. For example, in a zinc-copper galvanic cell, zinc undergoes oxidation at the anode, while copper ions are reduced at the cathode. This setup allows for the efficient conversion of chemical energy into electrical energy, which can then be harnessed for various applications, such as powering small devices in remote areas.Electrolytic Cells: Powering Industrial Processes
In contrast, electrolytic cells use electrical energy to drive non-spontaneous redox reactions, making them indispensable in industrial applications. This type of cell is essential for processes like electroplating, where a desired metal coating is applied to another metal by passing an electric current through an electrolyte solution. For example, in the electroplating of gold onto a jewelry piece, gold ions in the electrolytic bath are reduced and deposited onto the object’s surface, a process that wouldn’t occur naturally.Can galvanic cells be used in industrial processes?
While galvanic cells are primarily used for generating electrical energy from chemical reactions, their efficiency in small-scale, low-energy applications, like powering sensors or small electronic devices, is noteworthy.
What are the environmental impacts of using electrolytic cells?
Electrolytic cells, while powerful for industrial applications, require significant electrical energy inputs and can produce hazardous waste, such as spent electrolytes. Proper management and recycling practices are essential to mitigate environmental impacts.
Galvanic cells are inherently more efficient since they leverage natural redox reactions to generate electrical energy, while electrolytic cells demand a substantial input of external electrical energy to facilitate otherwise non-spontaneous reactions. Understanding these differences allows for better-informed decisions in various applications, from powering small, portable devices to large-scale industrial processes.
In conclusion, knowing the operational principles, efficiencies, and applications of both galvanic and electrolytic cells empowers professionals to select the most appropriate technology for their specific needs, ensuring optimal performance and sustainability. Whether your interest lies in sustainable energy generation or efficient industrial processes, these cells represent two sides of the same coin, each with its unique strengths and applications.