Hydrogen peroxide (H2O2) holds a vital place in various chemical processes and industrial applications, from disinfectants to rocket propellants. Understanding its Lewis structure is crucial for comprehending its chemical behavior and reactivity. This article delves into the nuances of the H2O2 Lewis structure, providing an expert perspective, practical insights, and actionable recommendations.
Introduction to the H2O2 Lewis Structure
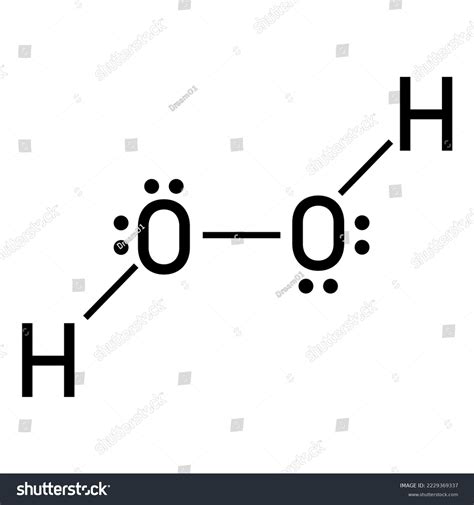
The H2O2 Lewis structure, representing the molecular arrangement of hydrogen peroxide, is pivotal for understanding its reactivity and applications. Hydrogen peroxide, composed of two hydrogen atoms and two oxygen atoms, exhibits an open book-like structure, where each hydrogen atom bonds with an oxygen atom, and these oxygens are further bonded together. This arrangement allows H2O2 to serve as both an oxidizing and reducing agent, which is instrumental in many industrial and biological processes.
Primary Insight: The Geometry and Bonding of H2O2
The H2O2 molecule presents a unique and insightful bonding configuration. Each hydrogen atom forms a single bond with its adjacent oxygen atom, resulting in two O-H bonds. The two oxygen atoms are then connected by a single bond, resulting in an open-book geometry. The molecule’s planar structure and bent shape are determined by the arrangement of the atoms around the central oxygen atoms. Understanding this planar geometry is critical for grasping H2O2’s role in redox reactions and its interaction with other molecules.
Technical Consideration: Electron Pairs and Resonance
In the Lewis structure of H2O2, the key technical aspect to consider is the distribution of lone pairs and the molecule’s resonance. Each oxygen atom not only forms a single bond with a hydrogen but also possesses two lone pairs of electrons. These lone pairs play a significant role in the molecule’s stability and reactivity. Resonance structures, though not common in H2O2, provide insight into the delocalization of electrons and enhance the understanding of the molecule’s electronic configuration. The balanced distribution of electrons and lone pairs within the H2O2 molecule aids in stabilizing its reactive nature, making it a potent chemical compound.
Key Insights
- Primary insight with practical relevance: Understanding H2O2’s open-book geometry elucidates its role in redox reactions.
- Technical consideration with clear application: Analyzing lone pairs and resonance offers deeper insight into the molecule’s reactivity and stability.
- Actionable recommendation: Utilize the Lewis structure of H2O2 to predict and analyze its behavior in various chemical reactions.
FAQ Section
What is the significance of H2O2’s planar geometry?
H2O2’s planar geometry is crucial as it influences the molecule’s reactivity, stability, and its ability to participate in redox reactions, impacting its practical applications in various fields.
Additional Considerations: Lone Pairs and Hydrogen Bonding
Beyond the basic Lewis structure, the presence of lone pairs on the oxygen atoms in H2O2 facilitates hydrogen bonding, which further influences the molecule’s physical properties. Hydrogen bonding between H2O2 molecules can affect its solubility in water, melting point, and boiling point. These properties are significant in its application as a bleaching agent and disinfectant, where its solubility and reactivity with other substances are crucial.
Understanding the H2O2 Lewis structure is not merely an academic exercise but a gateway to comprehending its wide range of applications. The structural configuration, electron distribution, and hydrogen bonding all intertwine to form a molecule that is as versatile as it is reactive. With this knowledge, one can leverage H2O2’s unique properties in various industrial and scientific endeavors.
By adhering to these insights, technical considerations, and actionable recommendations, the H2O2 Lewis structure becomes an invaluable tool for both educational and practical purposes.