Welcome to this comprehensive guide on understanding how many electrons sulfur holds. Sulfur, an essential element on the periodic table, plays a crucial role in various chemical reactions and processes. Whether you're a student, a chemist, or simply curious about the atomic structure of elements, this guide will walk you through everything you need to know about sulfur's electron count. We’ll begin by addressing common questions and challenges users face when learning about sulfur’s electrons.
One of the primary pain points learners often encounter is understanding the basic principles of atomic structure and electron configuration. Many struggle to grasp why elements like sulfur have specific electron arrangements and how this impacts their chemical behavior. This guide aims to demystify these concepts, providing step-by-step guidance with practical solutions, real-world examples, and expert advice to ensure you gain a solid understanding.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Look up the atomic number of sulfur, which is 16. This number directly corresponds to the number of electrons in a neutral sulfur atom.
- Essential tip with step-by-step guidance: To determine the electron configuration, start with the noble gas notation for sulfur, [Ne] 3s2 3p4. This means sulfur has two electrons in the 3s subshell and four in the 3p subshell.
- Common mistake to avoid with solution: Confusing protons with electrons. Remember, for a neutral atom, the number of protons equals the number of electrons. In the case of sulfur, both are 16.
Let’s dive deeper into understanding the electron count of sulfur and explore how to determine this for other elements.
Understanding Sulfur’s Electron Configuration
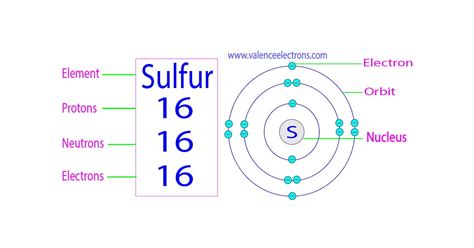
To fully comprehend how many electrons sulfur holds, it’s essential to first understand atomic structure. Atoms consist of a nucleus, which houses protons and neutrons, surrounded by electrons that travel in orbitals around the nucleus. Protons carry a positive charge, neutrons are neutral, and electrons possess a negative charge.
Each element on the periodic table is identified by its atomic number, which represents the number of protons in the nucleus of an atom. For sulfur, the atomic number is 16, which means a neutral sulfur atom has 16 protons and 16 electrons.
Electrons occupy various energy levels or shells, and each shell has subshells designated as s, p, d, and f. Electrons fill these subshells in a predictable order based on their energy levels. Sulfur’s electrons are distributed in the following way:
- The first shell (n=1) contains the 1s subshell with a maximum of 2 electrons.
- The second shell (n=2) includes the 2s and 2p subshells with a combined maximum of 8 electrons.
- The third shell (n=3) encompasses the 3s and 3p subshells with a maximum of 18 electrons.
For sulfur, the electron configuration is filled up to the 3p subshell:
Electron Configuration: 1s2 2s2 2p6 3s2 3p4
This means that sulfur has two electrons in the 1s subshell, two in the 2s subshell, six in the 2p subshell, two in the 3s subshell, and four in the 3p subshell.
To make this clearer, we can represent sulfur's electron configuration using noble gas notation. This method utilizes the nearest noble gas preceding the element and adds the remaining electron configuration after that noble gas:
Noble Gas Notation: [Ne] 3s2 3p4
Here, [Ne] stands for the electron configuration of neon, which is 1s2 2s2 2p6. We only need to list the remaining electrons, which for sulfur are 3s2 3p4.
Why Understanding Electron Configuration Matters
Grasping the electron configuration of elements like sulfur is essential for several reasons. It forms the foundation for understanding chemical bonding, reactivity, and the behavior of atoms in different compounds. By knowing the electron configuration, chemists can predict how sulfur will interact with other elements and form compounds, such as sulfates.
Moreover, a detailed understanding of electron configuration helps in comprehending the concepts of valence electrons, which determine how atoms bond together. Sulfur has six valence electrons (two in the 3s subshell and four in the 3p subshell), making it capable of forming multiple bonds and participating in a variety of chemical reactions.
Practical Examples of Sulfur’s Electron Configuration
To bring this concept to life, let’s explore some practical examples where understanding sulfur’s electron configuration is particularly useful:
Example 1: Formation of Sulfur Dioxide (SO2)
In the formation of sulfur dioxide, sulfur interacts with two oxygen atoms. Each oxygen atom needs two electrons to complete its outer shell and achieve a stable configuration similar to neon. Sulfur, with six valence electrons, can share two electrons with each oxygen atom:
- Each sulfur-oxygen bond involves sharing of electrons, forming double bonds.
- The resulting structure of SO2 has sulfur at the center, double-bonded to two oxygen atoms.
Example 2: Formation of Sulfur Trioxide (SO3)
When sulfur reacts with three oxygen atoms to form sulfur trioxide, the situation is slightly more complex:
- Sulfur can form three double bonds with three oxygen atoms.
- Due to resonance, the true structure involves a delocalized electron cloud across the sulfur and three oxygen atoms, providing additional stability to the molecule.
Example 3: Acid Rain and Sulfuric Acid (H2SO4)
Sulfur’s role in environmental chemistry, particularly in the formation of acid rain, is well-documented. When sulfur dioxide is released into the atmosphere and reacts with water vapor, it forms sulfuric acid:
- SO2 + H2O → H2SO3
- Further oxidation leads to H2SO4 (sulfuric acid).
- This acid rain can have detrimental effects on aquatic ecosystems and soil chemistry.
Understanding sulfur’s electron configuration allows you to predict these chemical reactions and their environmental impacts.
Practical FAQ
How do I determine the electron configuration of other elements?
To determine the electron configuration of any element, follow these steps:
- Find the atomic number of the element on the periodic table. This number indicates the number of protons and, for a neutral atom, the number of electrons.
- Use the Aufbau principle to fill the orbitals starting from the lowest energy level. The order is generally 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, and so on.
- Write the electron configuration in a concise form, indicating the number of electrons in each orbital. For example, for calcium (atomic number 20), the configuration is [Ar] 4s2.
- Alternatively, use noble gas notation for a more streamlined representation. Start with the nearest noble gas and add the remaining electrons. For calcium, it would be [Ar] 4s2.
Practice with