Understanding the relationship between milligrams (mg) and milliliters (ml) is fundamental in many fields, especially in pharmaceuticals and chemistry. Despite both units measuring quantities, mg measures mass while ml measures volume. This distinction is crucial to avoid confusion, especially when preparing solutions in labs or when dosing medication. This article aims to elucidate the difference and clarify the concept of ‘how many ml in a mg’.
The conversion between mg and ml is not straightforward and heavily depends on the density of the substance in question. To simplify, let’s start with the basic insight: one milliliter of water weighs exactly one gram at 4°C. However, this equivalency doesn’t apply universally.
Key Insights
- Mg measures mass, ml measures volume, so direct conversion isn’t possible without knowing the substance’s density.
- Density varies significantly among substances; water at 4°C is the exception where 1 ml = 1 g.
- Use concentration (e.g., mg/ml) for accurate conversions in practical applications.
Why Conversion Requires Substance Density
The relationship between mass and volume is defined by density, calculated as mass per unit volume. This property varies with temperature and substance. For example, water’s density at 20°C is approximately 0.9982 g/ml, meaning that 1 ml of water weighs less than 1 gram. In contrast, substances with higher density than water will weigh more than 1 gram per milliliter.Concentration as a Practical Approach
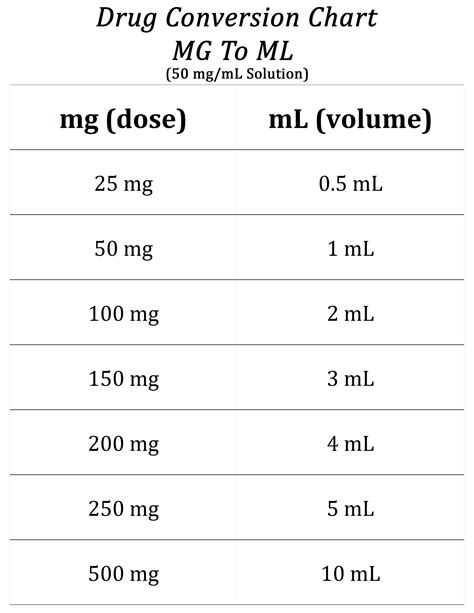
A more practical approach is understanding and using concentration measurements, such as mg/ml. This concentration indicates how many milligrams of a substance are present in one milliliter of solution. For instance, a 10 mg/ml solution means that each ml of the solution contains 10 mg of the active ingredient.In pharmaceutical settings, this is vital. For example, if a patient needs 50 mg of a drug, a 10 mg/ml solution would require 5 ml to meet the dosage requirement. This approach eliminates the need to worry about the density of the drug, focusing instead on the active ingredient concentration in the solution.
What if I need to convert between mg and ml for a specific substance?
You must first determine the density of the substance at the temperature you’re working with. Then, use the concentration formula (mg/ml) to convert. If you have a 2 g/ml density for a substance, 1 ml would weigh 2 mg.
Is it possible to convert directly between mg and ml without additional information?
No, because mg and ml measure different properties (mass and volume). Without knowing the substance’s density and how it behaves under specific conditions, a direct conversion isn't feasible.
Understanding the difference between mg and ml, along with the principle of density, is essential for anyone working in fields that require precise measurement and preparation of solutions. Always consider the substance’s density and use concentration to accurately convert between mg and ml in practical applications.