Understanding the concept of valence electrons is fundamental in chemistry, particularly in understanding the chemical behavior of elements like fluorine. As a key player in various scientific applications and industrial processes, fluorine’s electron configuration is crucial for both theoretical and practical chemistry. This article delves into the valence electrons of fluorine, providing expert insights with practical implications.
Understanding Electron Configuration
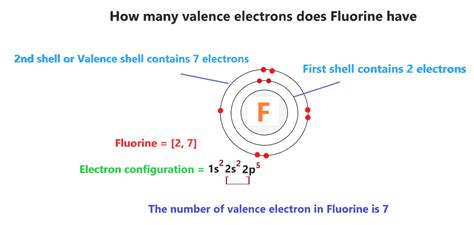
The journey to comprehend fluorine’s valence electrons begins with electron configuration. Electrons in an atom are arranged in shells around the nucleus, with each shell capable of holding a specific number of electrons. The arrangement follows the aufbau principle, where electrons fill orbitals starting from the lowest to the highest energy levels.
Fluorine, with an atomic number of 9, has nine electrons. The distribution is as follows: 2 electrons in the first shell, 7 in the second shell, and none in the third. The second shell, also known as the valence shell, holds seven electrons. These seven electrons in the second shell are the ones available for chemical reactions, making them the valence electrons.
The Role of Valence Electrons in Chemical Reactions
Valence electrons are pivotal in determining an atom’s chemical properties, as they participate in the formation of chemical bonds. Fluorine, being a highly reactive non-metal, seeks to achieve a stable electron configuration similar to neon, a noble gas. It achieves this by gaining one electron to fill its valence shell, forming a fluoride ion (F⁻). This characteristic makes fluorine an excellent candidate for forming ionic and covalent bonds, as seen in compounds like hydrogen fluoride (HF) and sodium fluoride (NaF).
Real-World Applications
Fluorine’s reactivity, driven by its valence electrons, finds applications in various fields. Fluorine is essential in the production of refrigerants, such as chlorofluorocarbons (CFCs), and in the synthesis of pharmaceuticals, particularly in the development of fluorinated drugs which often have enhanced metabolic stability and bioavailability.
Key Insights
- Primary insight with practical relevance: Fluorine has seven valence electrons which determine its high reactivity and its role in forming chemical bonds.
- Technical consideration with clear application: The electron configuration of fluorine explains its behavior in chemical reactions and its industrial applications.
- Actionable recommendation: For industrial chemistry, understanding fluorine's valence electrons can guide the synthesis of specific fluorinated compounds with desired properties.
FAQ Section
Why does fluorine have such high reactivity?
Fluorine’s high reactivity is due to its seven valence electrons, which it readily donates or shares to achieve a stable electron configuration. This tendency to gain an electron to complete its valence shell makes it highly reactive.
How does fluorine's valence electron configuration impact its use in pharmaceuticals?
Fluorine's unique valence electron configuration enables it to form stable yet highly reactive compounds. In pharmaceuticals, fluorine atoms can replace hydrogen atoms in drug molecules, often improving their effectiveness, metabolic stability, and ability to cross biological membranes.
This detailed exploration underscores the importance of understanding valence electrons in chemistry. Recognizing the specific number of valence electrons in elements like fluorine is not merely academic; it has profound implications in both scientific research and practical applications, from industrial chemistry to pharmaceuticals.