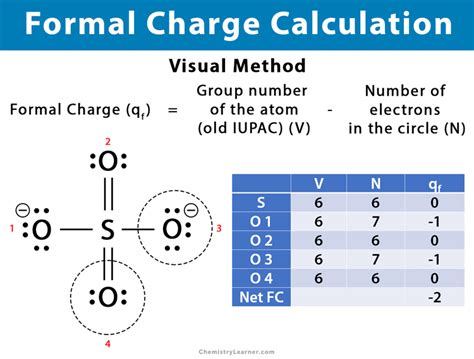
Calculating formal charge is a fundamental aspect of understanding molecular structures and predicting chemical behavior. The formal charge of an atom in a molecule provides insight into the distribution of electrons in the molecule, which is essential for drawing accurate Lewis structures and predicting chemical reactivity. This guide aims to demystify the concept of formal charge, offering practical insights and real-world examples for effective learning.
Key Insights
- Primary insight with practical relevance: Formal charge helps in determining the most accurate Lewis structure of a molecule.
- Technical consideration with clear application: Understanding formal charge allows for the prediction of molecular polarity and intermolecular forces.
- Actionable recommendation: Use the formal charge calculation as a diagnostic tool to verify the correctness of Lewis structures.
Understanding Formal Charge
The formal charge (FC) of an atom in a molecule is a hypothetical charge that represents how well an atom fulfills its electron requirements according to the octet rule. To calculate the formal charge, use the following formula:FC = (Valence electrons of the atom) – (Non-bonding electrons) – 0.5 × (Bonding electrons)
For example, in a molecule like carbon dioxide (CO₂), the carbon atom shares four electrons with the two oxygen atoms. The carbon atom starts with four valence electrons. It has no non-bonding electrons in this case and shares eight electrons in bonds (four bonds × two electrons per bond). Applying the formula:
FC for Carbon in CO₂ = 4 – 0 – 0.5 × 8 = 0
This calculation shows that the carbon atom in CO₂ has a formal charge of zero, indicating a balanced electron distribution.
Applying Formal Charge in Lewis Structures
When constructing Lewis structures, the goal is to achieve a structure where the formal charge is as minimized as possible, ideally zero, for each atom. This minimization aligns with the most stable electron configuration. For instance, in the case of ammonia (NH₃), the nitrogen atom forms three single bonds with hydrogen atoms and has one lone pair of electrons. The nitrogen atom has five valence electrons.Using the formula for nitrogen in NH₃:
FC for Nitrogen in NH₃ = 5 – 2 – 0.5 × 6 = -1
Although the nitrogen atom has a formal charge of -1, the overall formal charge distribution in NH₃ sums to zero, signifying a stable structure.
How does formal charge relate to molecular polarity?
Formal charge is closely related to molecular polarity because it affects the overall charge distribution within a molecule. A molecule with uneven formal charge distribution likely exhibits a dipole moment, leading to polar intermolecular forces.
Can formal charge ever be positive?
Yes, formal charge can be positive or negative. A positive formal charge indicates that an atom has fewer electrons than it should ideally hold, while a negative formal charge indicates it has more electrons.
In conclusion, calculating formal charge is a powerful tool in chemistry that aids in determining accurate Lewis structures and predicting molecular behavior. By understanding and applying the principles of formal charge, chemists can better predict molecular interactions, reactivity, and stability. Through careful calculation and analysis, formal charge becomes an indispensable element in the arsenal of molecular science.