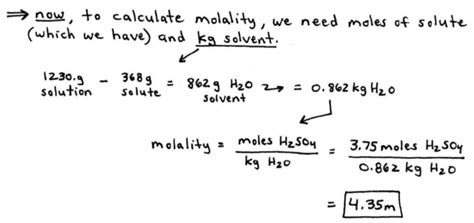The concept of molality is critical in various scientific disciplines, particularly in chemistry and biochemistry, where precise measurement of solute concentration in a solution can significantly affect experimental outcomes. Unlike molarity, which relies on the volume of the solution, molality is based on the mass of the solvent. This difference can be pivotal in research settings where density changes with temperature must be accounted for. Here’s a focused exploration of how to master molality calculations with practical, evidence-based insights.
Key Insights
- Understanding molality's base on solvent mass leads to greater accuracy in temperature-sensitive experiments.
- Precision in measurement tools and careful calculation steps ensure reliable results.
- Use of a calibrated balance and volume measurements of solvent will enhance accuracy.
Molality is defined as the number of moles of solute per kilogram of solvent. This measurement unit offers significant advantages in scenarios where volume might fluctuate due to thermal expansion or compression. Here’s a comprehensive guide to calculating molality.
Steps to Calculate Molality
The formula for molality (m) is straightforward:
m = moles of solute / kilograms of solvent
Follow these steps for an accurate calculation:
- Weigh the Solvent: Use a calibrated analytical balance to measure the exact mass of the solvent in kilograms. Accuracy here is crucial since even minor deviations can skew results.
- Determine the Moles of Solute: To find the moles of solute, divide the mass of the solute by its molar mass (molecular weight in grams per mole). This conversion is essential as it links the mass measurement with the chemical quantity.
- Calculate Molality: With the moles of solute and the mass of the solvent, plug these values into the molality formula. A clear and careful calculation will yield precise molality.
Applications in Chemical Experiments
In chemical laboratories, molality is often employed in precise experiments where temperature variations are expected. For instance, in colligative properties studies such as boiling point elevation or freezing point depression, molality offers a more stable measure. Research on solutions with ionic solutes, like salts in water, benefits from using molality since these solutions can expand or contract upon dissolution. Here, accurate molality calculations ensure the integrity of the experimental data, contributing to reliable and reproducible results.
Why is molality preferred over molarity in certain experiments?
Molality is favored in experiments where solvent volume might change with temperature, providing a more consistent measure of solute concentration.
What’s the main difference between molality and molarity?
Molality is based on the mass of the solvent while molarity is based on the volume of the solution, affecting their relevance in different experimental conditions.
This detailed guide and understanding of molality will empower you to conduct precise and reliable chemical analyses, ensuring that your results are accurate and valuable to your scientific research. Precision in every step, from weighing solvents to calculating the final molality, is key to mastering this essential concept.