In the realm of chemistry, the concept of atomic radius is pivotal for understanding chemical bonding, molecular structures, and periodic trends. Grasping the atomic radius can be challenging, but with the right guidance, it becomes a manageable concept for anyone. This guide offers practical, user-focused steps to help you easily find and comprehend atomic radius. We’ll walk through real-world examples, problem-solving tips, and ensure that you gain actionable knowledge to build your understanding progressively.
Understanding Atomic Radius: Your First Step
Atomic radius is a measure of the size of an atom and is crucial in determining how atoms interact with each other. Whether you’re a student grappling with periodic table trends or a professional navigating complex chemical structures, understanding atomic radius is essential. Misconceptions around its calculation and significance can lead to confusion, but this guide aims to clear those up, providing you with easy-to-follow strategies to master this concept.
Quick Reference Guide
Quick Reference
- Immediate action item: Identify elements you need to determine atomic radius for
- Essential tip: Use the periodic table to locate atomic radius data
- Common mistake to avoid: Confusing atomic radius with ionic radius
Let's dive deeper into actionable steps to find and understand atomic radius. Here, we'll break down complex information into simple, practical actions.
How to Find Atomic Radius: Basic Steps
Finding the atomic radius starts with understanding its basic definition. Atomic radius refers to the distance from the center of an atom’s nucleus to the outer boundary of its electron cloud. Here’s how to proceed:
Step 1: Identify the Element
Start by identifying the element for which you need the atomic radius. You can find elements on the periodic table.
Step 2: Locate the Atomic Radius Data
There are several ways to find atomic radius data:
- Refer to an authoritative source like the National Institute of Standards and Technology (NIST).
- Use educational chemistry websites that provide periodic tables with atomic radius data.
- Consult chemistry textbooks that often include periodic tables with atomic radius values.
For instance, the atomic radius of carbon is approximately 77 picometers (pm).
Step 3: Understand Units and Trends
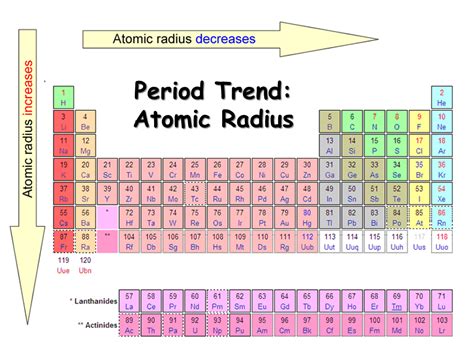
Atomic radius is typically measured in picometers (pm) or angstroms (Å). It’s crucial to understand the trends in atomic radius:
- Down a group on the periodic table, atomic radius increases due to the addition of electron shells.
- Across a period, atomic radius generally decreases because of the increased effective nuclear charge, pulling electrons closer to the nucleus.
Example: Consider elements in Group 1 (alkali metals) where atomic radius increases significantly from lithium (Li) to cesium (Cs) as you move down the group.
Detailed Insights on Atomic Radius Calculation
Calculations involving atomic radius can seem daunting, but they’re rooted in fundamental principles that become easier with practice.
Understanding Different Types of Atomic Radius
There are several types of atomic radius, including:
- Covalent radius: Half the distance between the nuclei of two identical atoms bonded covalently.
- Ionic radius: The size of an ion, either cation or anion. Note that this differs from atomic radius.
- Van der Waals radius: The distance between two similar atoms, with each atom having a non-bonded electron cloud.
For instance, the covalent radius of hydrogen is around 31 pm, reflecting the size of the hydrogen atom when it forms covalent bonds.
Step-by-Step Calculation Process
To calculate the covalent radius, follow these steps:
- Identify the element and determine its position on the periodic table.
- Use a reference source to find the typical covalent radius value.
- Apply this value in understanding chemical bond formations.
Example calculation for chlorine:
Using a reference table, the covalent radius of chlorine (Cl) is approximately 99 pm. This measurement is crucial when considering how chlorine atoms interact in diatomic chlorine (Cl₂) or when bonding with other elements.
Applying Atomic Radius in Chemistry
Understanding atomic radius is essential for applications in chemistry:
- Predicting chemical reactivity by considering how atom sizes affect bond lengths.
- Determining molecular geometry and the shape of molecules based on atomic sizes.
- Understanding solubility and polarity through the lens of atomic radius differences.
For instance, larger atomic radii in Group 1 elements lead to weaker bonds and greater reactivity with nonmetals.
Practical FAQ Section
What’s the difference between atomic radius and ionic radius?
The atomic radius measures the size of a neutral atom, while the ionic radius measures the size of an ion after it has gained or lost electrons. Ionic radius varies significantly from atomic radius due to changes in electron configuration when an atom becomes an ion. For example, a sodium atom (Na) has an atomic radius of about 186 pm, but a sodium ion (Na⁺) has an ionic radius of around 95 pm because it loses an electron, pulling the remaining electrons closer to the nucleus.
Why does atomic radius decrease across a period?
Across a period, atomic radius decreases because of the increasing nuclear charge. As you move from left to right across a period, additional protons are added to the nucleus, resulting in a greater positive charge that pulls electrons closer to the nucleus. This reduced electron cloud size results in a smaller atomic radius.
How can I find the atomic radius of transition metals?
Transition metals can be a bit tricky due to their complex electron configurations. The atomic radius of transition metals varies within a period due to the lanthanide and actinide contraction. To find accurate atomic radius data, refer to specialized chemistry databases or textbooks. For instance, the atomic radius of iron (Fe) is about 126 pm.
Mastering the concept of atomic radius not only aids in understanding basic chemical principles but also paves the way for comprehending more advanced topics in chemistry. By following the practical steps and tips outlined here, you can easily find and understand atomic radius, leading to better comprehension of chemical behaviors and reactions.