Neutrons are fundamental subatomic particles essential to our understanding of atomic nuclei and nuclear physics. They are neutral particles that contribute to the stability of an atom’s nucleus alongside protons. This article will provide a straightforward, expert-led exploration into the basics of finding neutrons, ensuring a grasp of their relevance through evidence-based insights and real-world applications.
Key insights box:
Key Insights
- Neutrons play a pivotal role in nuclear stability, influencing atomic mass.
- Isotopes are differentiated by their neutron counts, impacting scientific and industrial applications.
- Neutron detection instruments like scintillators and Geiger counters are essential tools for their identification.
Understanding neutrons begins with their role within an atom’s nucleus. In a neutral atom, the number of protons equals the number of electrons. Neutrons, which possess no charge, are crucial for binding protons together to form the nucleus. This binding is essential for atomic stability and mass, as the sum of protons and neutrons (nucleons) determines the atomic mass number.
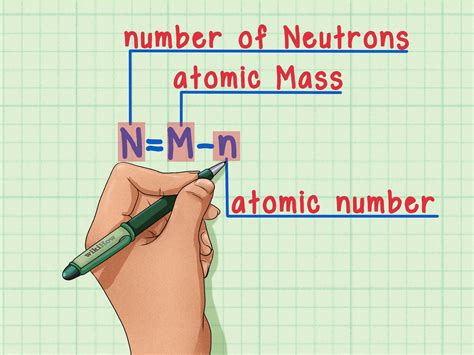
To find neutrons, it’s essential to know the atomic number (Z), which denotes the number of protons, and the atomic mass number (A), which is the total number of protons and neutrons. The equation to determine the number of neutrons (N) is straightforward: N = A – Z. For example, carbon-12 (C-12) has an atomic mass number of 12 and an atomic number of 6, meaning it has 12 – 6 = 6 neutrons.
The significance of neutrons extends beyond basic atomic structure into various scientific and industrial domains. Isotopes, atoms of the same element with different neutron counts, exhibit different physical and chemical properties, crucial for applications like medical imaging and treatment. For instance, carbon-14 (with 8 neutrons) is utilized in radiocarbon dating, while isotopes like uranium-235 are pivotal in nuclear reactors.
To find neutrons practically, scientists use sophisticated instruments that detect these neutral particles. A scintillator, for instance, is a material that emits light upon neutron interaction, making it easy to count and analyze. Similarly, Geiger counters provide real-time detection and quantification of neutron radiation levels, ensuring safety in nuclear facilities and research environments.
When investigating neutron interactions, consider these critical factors: 1. Isotopic Identification: Different isotopes have distinct neutron counts, which influence their stability and use in various applications. 2. Detection Methods: Use advanced technologies like scintillators and Geiger counters for precise neutron identification and measurement. 3. Nuclear Reactions: Understanding nuclear reactions that involve neutron capture or emission can provide deeper insights into neutron behavior and applications.
FAQ section:
What is the best way to detect neutrons?
The best way to detect neutrons involves using scintillators, which are highly sensitive materials that emit light upon neutron interaction, making it easy to count and analyze the neutrons. Additionally, Geiger counters offer real-time detection and quantification of neutron radiation levels.
Why are neutrons important in nuclear power plants?
Neutrons are crucial in nuclear power plants because they facilitate the fission process. In uranium-235, neutrons are absorbed to split the nucleus, releasing energy that generates electricity. Without a controlled supply of neutrons, the nuclear chain reaction cannot proceed, making neutron control vital for safe and effective power generation.
Understanding neutrons, their role in atomic structure, and their detection methods not only enriches scientific knowledge but also drives technological advancements across multiple fields. With a focus on practical applications and real-world examples, this guide demystifies the pursuit of finding neutrons.