Pinning down whether sugar truly is a compound can ignite a fascinating debate in both the culinary and scientific worlds. For centuries, our understanding of sugar has evolved from a simple sweetener to a complex molecule. To unravel this, let’s dive deep into the nuances of what sugar represents in both everyday use and scientific context.
Key insights box:
Key Insights
- Sugar is technically classified as a compound due to its chemical composition.
- Its molecular structure reveals a precise arrangement of atoms, making it fit within chemical definitions.
- Understanding this molecular basis has practical implications in fields like nutrition and biochemistry.
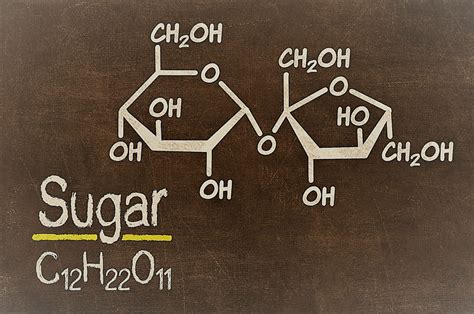
Understanding sugar from a chemical perspective involves examining its molecular composition. Most commonly, we think of sugar as table sugar or sucrose. Sucrose is a disaccharide, meaning it is composed of two monosaccharides: glucose and fructose. When we delve deeper, we observe that this intricate assembly of atoms occurs through a condensation reaction, where water is removed, creating a stable sugar molecule with the formula C₁₂H₂₂O₁₁.
This chemical composition places sugar in the broader category of carbohydrates. Carbohydrates are macromolecules that consist of carbon, hydrogen, and oxygen atoms. The precise ratios and the arrangement of these atoms are what define sugar as a compound. The complexity doesn’t end here; different forms of sugar like glucose, fructose, and lactose, have their unique molecular structures, all of which solidify their classification as compounds in chemistry.
The Chemical Structure of Sugar
Sucrose, as previously mentioned, is made up of glucose and fructose units bonded together. When these sugars are enzymatically broken down in the digestive system, they revert to their monosaccharide forms, indicating the specific molecular bonds that define sucrose. This breakdown is essential for our understanding of sugar metabolism in the body and highlights its role in providing quick energy.
Another critical aspect of sugar’s chemical structure is its crystalline form. In its pure state, sugar forms distinct crystals, a direct result of the orderly arrangement of its molecules. These crystals exhibit a specific melting point, further distinguishing sugar from a mixture or solution. When we examine these properties under a microscope, the clarity in which the molecules align reveals the true nature of sugar as a compound.
The Role of Sugar in Biochemistry
From a biochemical standpoint, sugar plays pivotal roles in various metabolic pathways. One of the primary functions is energy production. Once broken down, glucose is fed into the glycolytic pathway and subsequently the citric acid cycle to generate ATP, the energy currency of the cell. This process underscores the significance of understanding sugar’s chemical nature.
Additionally, sugar is integral in cellular communication and signaling. Molecules like sialic acid (a derivative of glucose) are key components in the synthesis of glycoproteins and glycolipids. These molecules are crucial in cell recognition, immune response, and tissue development, emphasizing the multifaceted role sugar plays beyond mere sweetness.
FAQ section:
Is sugar the same as glucose?
No, while both are carbohydrates, sugar typically refers to sucrose, a disaccharide composed of glucose and fructose. Glucose is a monosaccharide that is a fundamental energy source for cells.
Why is understanding sugar’s chemical structure important?
Understanding the chemical structure of sugar is crucial for nutritional science, metabolism, and biochemical processes. It helps in formulating dietary guidelines and developing treatments for metabolic disorders.
In conclusion, sugar transcends its sweet taste to embody a complex molecule with significant scientific and practical implications. Its classification as a compound stems from its precise molecular composition and structural integrity, which are fundamental to its biochemical roles. Whether viewed through the lens of everyday use or scientific rigor, the true nature of sugar as a compound becomes evident.