Is Water a Pure Substance Debate
Understanding whether water is a pure substance is a question that often perplexes students and even seasoned scientists at times. To unravel this debate, it’s essential to grasp the fundamental concepts of chemistry and delve into the intricate nature of water. This guide will help demystify the complexities around the purity of water, offering a clear and practical path to understanding. Whether you’re a student navigating your first chemistry class or an adult seeking to deepen your scientific knowledge, this guide provides actionable insights and real-world examples to ensure you grasp the nuances involved.
Problem-Solution Opening Addressing User Needs
Many people, especially students and casual learners, often struggle with the question: “Is water a pure substance?” This dilemma arises because while water (H₂O) seems straightforward, its practical applications involve complex interactions with various substances. The core problem lies in distinguishing between pure substances and mixtures. Understanding this distinction is crucial for anyone involved in scientific research, environmental science, or even daily life, where water’s role as a solvent and reactant cannot be overstated. This guide aims to break down the debate with clear, actionable advice and real-world examples, offering a comprehensive overview of why and how water can sometimes be viewed as both a pure substance and a complex mixture.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always check the source and purity level of water used in scientific experiments.
- Essential tip with step-by-step guidance: Use distilled water for experiments requiring pure H₂O to avoid contamination from minerals and impurities.
- Common mistake to avoid with solution: Mislabeling tap water as pure due to its molecular composition without considering the dissolved substances.
What Makes a Substance ‘Pure’?
To determine if water is a pure substance, we must first understand what it means to be ‘pure.’ In chemistry, a pure substance has a uniform and definite composition. It consists of a single type of particle, whether it’s an atom, molecule, or compound. A pure substance maintains consistent properties throughout.
When we refer to a pure substance in this context, we're considering only H₂O molecules, devoid of any other elements or compounds mixed within. However, the real world seldom offers substances in this pure form due to natural contaminations and impurities.
Detailed Explanation: Is Water a Pure Substance?
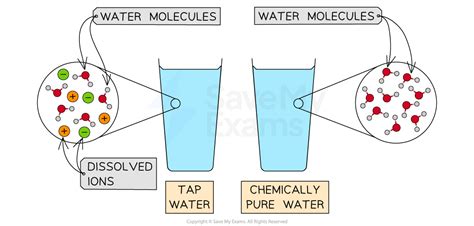
The simplicity of water’s chemical formula (H₂O) belies its complex nature in practical settings. In an ideal laboratory scenario, water can and should be considered a pure substance if it is devoid of any impurities. However, natural water sources often contain various dissolved substances such as minerals, salts, and organic materials. This is where the debate often arises.
Pure Water: The Ideal
Pure water, also known as distilled water, is obtained by boiling water to create steam and then condensing that steam back into a liquid. This process removes virtually all impurities, including salts, minerals, and microorganisms, leaving behind only H₂O molecules. In a lab setting, distilled water is the standard for ensuring reactions occur only based on the properties of H₂O.
Natural Water: A Mixture
On the other hand, water found in nature, such as lakes, rivers, and even groundwater, is inherently a mixture. This natural water contains dissolved gases like carbon dioxide, organic matter, and a myriad of dissolved salts. Even the purest natural water will have trace amounts of various substances.
Here's a practical example: when pure water is used in a chemistry lab to synthesize another substance, it ensures that no other variables interfere with the experiment. Conversely, using natural water could introduce unwanted variables, complicating results and interpretations.
Industrial vs. Laboratory Standards
Different industries have different standards for what they consider ‘pure’ water. For example, pharmaceuticals might use ultra-pure water (UPW) for manufacturing, requiring rigorous purification processes to meet strict purity criteria. These processes might include reverse osmosis, ion exchange, and additional distillation to achieve the desired purity level.
In contrast, laboratory-grade water might simply be distilled or deionized but doesn't necessarily need to meet the stringent purity standards of the pharmaceutical industry.
How to Determine the Purity of Water
If you’re interested in determining the purity of water in a practical setting, here’s a step-by-step guide to ensure you’re using the purest form possible for your needs:
- Collect water samples from various sources.
- Test for specific contaminants using appropriate kits or lab equipment. Common tests include:
- pH tests
- Turbidity tests
- Contaminant-specific tests (for heavy metals, microbial content, etc.)
- Use distillation or reverse osmosis if necessary to purify the water.
- Confirm the purity by retesting the water post-purification.
Step-by-Step Purification Process
To purify water to a level where it can be considered a pure substance, follow these steps:
1. Boiling and Condensation: Simple distillation is effective in removing most impurities. Here’s how:
- Heat the water in a container until it boils, converting to steam.
- Capture the steam and allow it to condense back into a liquid in a clean, cold container.
- The condensed water is distilled and much purer than the original.
2. Reverse Osmosis: For more complex impurities:
- Water passes through a semi-permeable membrane under pressure.
- The membrane filters out most contaminants, including salts and other dissolved substances.
- The purified water exits on one side, while impurities are left behind.
3. Deionization: To remove specific ions:
- Pass the water through an ion exchange resin that binds to unwanted ions.
- The clean water emerges devoid of ionic contaminants.
Practical FAQ
What are the different types of impurities found in natural water?
Natural water often contains various types of impurities, including:
- Minerals: Calcium, magnesium, sodium, potassium, etc.
- Organic matter: Decomposed plants and animals, organic chemicals.
- Microorganisms: Bacteria, viruses, protozoa.
- Gases: Carbon dioxide, oxygen, nitrogen.
- Inorganic chemicals: Chlorides, sulfates, phosphates.
Understanding these impurities helps in deciding the level of purification needed for specific uses.
If you have further questions or need more specific details on water purity, these points should guide you in understanding and ensuring the purity of water for your specific needs.