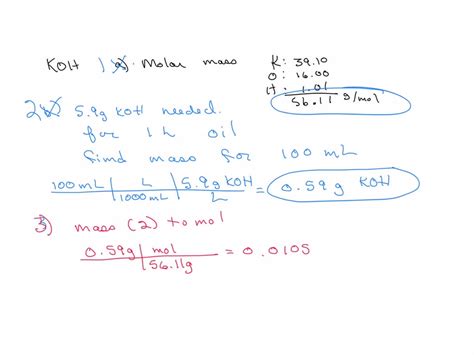Understanding Koh Molar Mass: An Essential Chemistry Insight
If you’re venturing into the world of chemistry, understanding molar mass is an essential skill. Knowing the molar mass of substances like potassium hydroxide (KOH) helps in a variety of practical scenarios, from lab calculations to everyday chemical applications. In this guide, we’ll provide a detailed and practical approach to understanding how to calculate and utilize the molar mass of KOH, ensuring you’re equipped with both the knowledge and the skills to implement these insights seamlessly in your studies or professional work.
The Problem-Solution Opening
Struggling to grasp the concept of molar mass or unsure how to apply it to KOH? Many students find chemistry challenging due to the abstract nature of these concepts, which often seems disconnected from real-world applications. However, once you understand how to calculate molar mass, you’ll find that it becomes a powerful tool for any chemistry enthusiast or professional. This guide will break down the concept into digestible, practical steps, providing you with actionable advice to overcome these challenges and apply these principles effectively.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Use the periodic table to find atomic masses of K, O, and H.
- Essential tip with step-by-step guidance: Add up the atomic masses to calculate the molar mass of KOH.
- Common mistake to avoid with solution: Ensure each element's atomic mass is used accurately.
How to Calculate Molar Mass of KOH
Calculating the molar mass of KOH is straightforward once you understand how to use the atomic masses of its constituent elements. Follow these steps:
Step 1: Gather Atomic Masses
First, find the atomic masses of potassium (K), oxygen (O), and hydrogen (H). These values can be found on the periodic table:
- Potassium (K): approximately 39.10 g/mol
- Oxygen (O): approximately 16.00 g/mol
- Hydrogen (H): approximately 1.01 g/mol
Step 2: Add the Atomic Masses Together
To find the molar mass of KOH, sum the atomic masses of each element:
Molar mass of KOH = (39.10 g/mol) + (16.00 g/mol) + (1.01 g/mol) = 56.11 g/mol
Step 3: Verify Your Calculation
Double-check your work by ensuring each atomic mass was added correctly and that your final calculation aligns with the molar mass information found in chemical references or reliable sources.
Here’s a practical tip: keep a handy periodic table for quick reference when you need atomic masses. Some digital tools or smartphone apps can also provide these values instantly.
Detailed How-To Section: Practical Examples
Now that you’ve got the basics down, let’s delve deeper with practical examples. These scenarios will illustrate how to apply molar mass calculations in real-world settings, offering you a clearer and more practical understanding.
Example 1: Determining the Number of Moles
Imagine you have a 112.22 gram sample of KOH. To determine how many moles this corresponds to, use the formula:
Number of moles = mass (g) / molar mass (g/mol)
Substitute the given values:
Number of moles = 112.22 g / 56.11 g/mol = 2.00 moles
Example 2: Converting to Grams
You have 0.5 moles of KOH and need to find its mass. Use the formula:
mass (g) = number of moles * molar mass (g/mol)
Substitute the given values:
mass (g) = 0.5 moles * 56.11 g/mol = 28.055 g
Example 3: Preparation of a KOH Solution
Suppose you need to prepare 250 mL of a 2 M KOH solution. To do this, you need to calculate the mass of KOH required. Start by calculating the number of moles needed:
Number of moles = molarity (M) * volume (L)
First, convert 250 mL to liters:
250 mL = 0.25 L
Now calculate the number of moles:
Number of moles = 2 M * 0.25 L = 0.5 moles
Now find the mass:
mass (g) = number of moles * molar mass (g/mol)
mass (g) = 0.5 moles * 56.11 g/mol = 28.055 g
Therefore, you need 28.055 grams of KOH to prepare the solution.
Practical FAQ
Why is molar mass important in chemical reactions?
Molar mass is crucial because it allows you to convert between the mass of a substance and the amount in moles, which is essential for stoichiometric calculations. This helps in determining the quantities of reactants needed or the yields of products in chemical reactions.
For example, if you know the molar mass of KOH, you can accurately determine how much KOH to use in a reaction to achieve the desired stoichiometric ratio.
By mastering molar mass calculations, you unlock a deeper understanding of chemical processes, enabling you to tackle complex problems with confidence and precision. With these examples and insights, you’ll gain the skills needed to apply molar mass calculations in both academic and professional settings.
Remember, practice makes perfect. The more you work through these calculations, the more intuitive they will become. Don’t hesitate to use online resources, textbooks, or consult with instructors to deepen your understanding and solve any lingering questions. With this knowledge, you’re well-equipped to take your chemistry endeavors to the next level.