Understanding methane’s Lewis structure is fundamental to grasping the basics of molecular chemistry and how atoms bond together. Many beginners find this topic daunting because it blends concepts of valence electrons, bonding, and molecular geometry. Worry not! This guide is crafted to take you through each step with actionable advice, real-world examples, and practical solutions that demystify the process. By the end of this guide, you’ll have a solid grasp of methane’s Lewis structure and how it plays into larger chemical concepts.
Why Understanding Methane’s Lewis Structure is Crucial
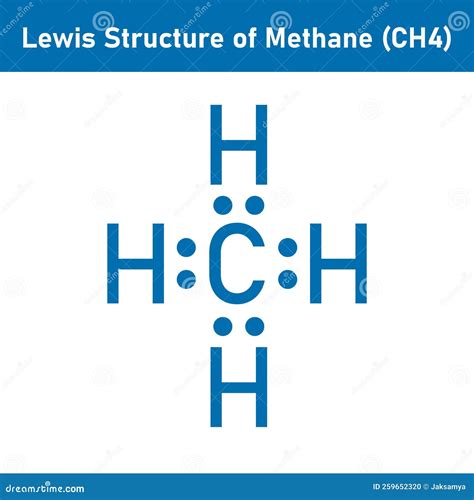
The methane molecule (CH₄) is the simplest hydrocarbon and serves as a cornerstone in learning organic and inorganic chemistry. Understanding its Lewis structure—a way of representing how atoms bond together—gives you a foundational skill for predicting the properties and reactivity of many more complex molecules. Methane’s structure is straightforward, which makes it an excellent entry point for grasping the complexities of Lewis structures.
Quick Reference Guide
Quick Reference
- Immediate action item: Draw a rough sketch of carbon and hydrogen atoms.
- Essential tip: Remember that carbon has four valence electrons and hydrogen has one.
- Common mistake to avoid: Forgetting to distribute electrons equally around the central atom.
Step-by-Step Guide to Drawing Methane’s Lewis Structure
Let’s break down the process of creating methane’s Lewis structure into manageable steps. We’ll start from the basics and gradually build up to the full structure.
Step 1: Understanding Valence Electrons
First, let’s talk about valence electrons. These are the outermost electrons of an atom and play a crucial role in chemical bonding. For carbon, located in group 14 of the periodic table, it has four valence electrons. Hydrogen, in group 1, has one valence electron. Knowing this will help you visualize how many bonds they can form.
Step 2: Calculating Total Valence Electrons
To draw the Lewis structure, start by calculating the total valence electrons. Methane has one carbon atom and four hydrogen atoms. Carbon contributes 4 valence electrons and each hydrogen contributes 1, leading to a total of 8 valence electrons.
Step 3: Drawing the Central Atom
Begin with the central atom, which is carbon in methane. Place it in the center of your sketch.
Step 4: Bonding Hydrogens to Carbon
Each hydrogen atom needs two electrons to achieve a full outer shell (just like helium). Carbon can share its four valence electrons to form four bonds with four hydrogen atoms, each forming a single covalent bond. This allows each hydrogen to fulfill its electron requirement while using up all of carbon’s valence electrons.
Step 5: Distributing Remaining Electrons
Since carbon needs a full octet and each hydrogen needs two electrons, this bonding method perfectly meets both atoms’ requirements. No lone pairs are needed because all valence electrons are used in bonding.
Step 6: Double-Check Electron Usage
To ensure correctness, count all the electrons. Carbon shares its four valence electrons, and each hydrogen shares one electron, totaling eight electrons, which matches our earlier calculation.
Step 7: Drawing the Lewis Structure
Here’s how you write the final Lewis structure:
- Place the carbon atom in the center.
- Draw four single bonds from the carbon to four hydrogen atoms.
- Each hydrogen should have two dots (representing the shared electron pair) around it.
Final Lewis Structure: H | H-C-H | H
Practical FAQ
Why is it important to balance the Lewis structure?
Ensuring that the Lewis structure is balanced is crucial for accurately representing the molecule’s chemical properties. Every atom should achieve the electron configuration of the nearest noble gas. For carbon, it means achieving a complete octet, and for hydrogen, it means having two electrons. Balancing your structure ensures that you have correctly accounted for the molecule’s electrons and that it’s stable and accurate.
Common Pitfalls and Solutions
When drawing Lewis structures, it’s easy to fall into common traps. Here are a few mistakes and how to avoid them:
- Mistake: Failing to use all valence electrons. Solution: Always ensure you have used all valence electrons before finalizing your structure.
- Mistake: Misplacing electrons, creating an incorrect bonding picture. Solution: Carefully count electrons each time you form a bond or place a lone pair.
- Mistake: Drawing hydrogen atoms incorrectly bonded to the central atom. Solution: Hydrogen atoms should always form single bonds in organic compounds like methane.
Best Practices for Learning Lewis Structures
To master Lewis structures, practice is key. Here are some best practices that will help you become proficient:
- Practice drawing simple molecules like methane before moving on to more complex ones.
- Use visual aids like diagrams and online tools to help solidify your understanding.
- Review and rework older problems to strengthen your muscle memory.
- Seek out additional resources like educational videos, books, or interactive tutorials.
By following these steps and best practices, you’ll have a robust understanding of methane’s Lewis structure. This foundational knowledge will pave the way for mastering more complex molecules and chemical concepts. Remember, practice and repetition are your best allies in chemistry. Happy studying!