Precision is paramount in chemistry. Whether quantifying substances, understanding molecular interactions, or predicting reaction outcomes, accurate conversion from moles to atoms is essential. This guide delves into expert perspectives, practical insights, and evidence-based strategies to navigate the complex landscape of mole-to-atom conversions, ensuring your chemical endeavors are precise and effective.
Key Insights
- Understanding Avogadro’s number is critical for mole-to-atom conversions.
- Precision in measurements ensures accurate chemical reactions.
- Utilize reliable conversion tools and software for better results.
Understanding Avogadro’s Number
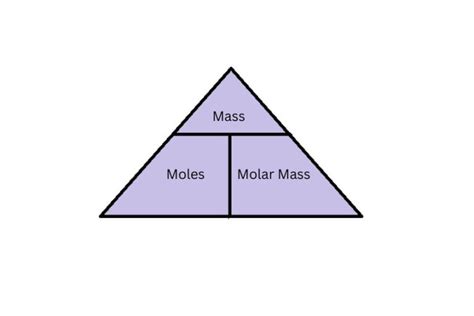
Avogadro’s number, approximately 6.022 x 10^23, is the cornerstone of mole-to-atom conversions. It represents the number of atoms, molecules, or particles in one mole of a substance. This constant is integral to the mole concept, which is foundational to chemistry. For example, one mole of carbon-12 atoms contains exactly 6.022 x 10^23 carbon atoms. Using this principle, chemists can convert between moles and atoms, ensuring precise stoichiometry in chemical reactions.Precision in Measurements
Accurate measurement tools are crucial for precision in chemistry. High-precision balances and volumetric flasks minimize errors, ensuring that the mole calculations translate into accurate atomic counts. For instance, when working with a 10.00g sample of sodium chloride, a precise balance is required to determine its exact molar mass, aiding in the conversion to moles and then to atoms. This meticulous approach is not just about numbers but about the reliability and reproducibility of results, which is crucial in both laboratory and industrial settings.When executing chemical reactions, precision in measurement translates into successful outcomes. Whether titrating an acid with a base or synthesizing a complex molecule, the ability to accurately measure moles and convert them to atoms is vital. This precision is not just about achieving the correct product; it’s about ensuring safety and efficacy in all chemical processes.
How do I ensure my measurements are precise?
Use calibrated instruments, follow standard protocols for measurement, and cross-verify with known standards to ensure accuracy.
Can I use online tools for mole-to-atom conversions?
Yes, reliable online calculators and software tools can assist with conversions, but always double-check with manual calculations for verification.
In the realm of chemistry, where precision dictates success, mastering the conversion from moles to atoms through an understanding of Avogadro’s number and employing precise measurement techniques is essential. By adhering to these principles, chemists can achieve the accuracy necessary for their experiments and reactions, thereby advancing scientific knowledge and innovation. This guide underscores the importance of these foundational concepts in achieving precision and success in chemical endeavors.