Navigating the labyrinth of IUPAC (International Union of Pure and Applied Chemistry) compound naming can feel overwhelming, especially when aiming for precision in scientific communication. This guide is designed to unravel these complexities, providing you with step-by-step guidance, practical solutions, and a wealth of tips to master IUPAC compound naming. By the end of this guide, you will not only understand the principles of IUPAC naming but also know how to apply them effectively to ensure clarity and accuracy in your scientific writing.
Understanding IUPAC Compound Naming: A Problem-Solution Approach
Understanding IUPAC compound naming is crucial for scientists to communicate the structures of chemical compounds unambiguously. Misnaming can lead to misunderstandings and errors in scientific research. The IUPAC system provides a systematic method to name compounds based on a set of rules, ensuring that each name corresponds to a unique structure. This guide will walk you through the essentials of these rules, providing actionable advice to ensure you can name compounds accurately and confidently.
Quick Reference: Essentials of IUPAC Naming
Quick Reference
- Immediate action item with clear benefit: Always start by identifying the longest carbon chain in organic compounds to determine the base name.
- Essential tip with step-by-step guidance: For organic compounds, use prefixes to denote substituents and numbers to indicate their positions.
- Common mistake to avoid with solution: Avoid using common names (like ethyl alcohol for ethanol) unless specified, as IUPAC names are preferred for precision.
Step-by-Step Guide to Naming Inorganic Compounds
Inorganic compound naming follows different principles compared to organic compounds. Here’s a detailed step-by-step guide to mastering the basics of IUPAC naming for inorganic compounds:
Identify the Cation and Anion
In inorganic compounds, you start by identifying the cation (positive ion) and anion (negative ion). The cation is named first, followed by the anion. For instance, in NaCl, sodium (Na+) is the cation and chloride (Cl-) is the anion. The name of the compound is formed by combining these names.
Name Simple Acids
For acids containing hydrogen and one other element, the name is derived from the element’s name with an ‘ic’ or ‘ous’ ending depending on the oxidation state of the element. For example, hydrochloric acid (HCl) is named using the element’s name, chlorine, and the acid suffix ‘acid’.
Use Stock Nomenclature for Transition Metals
Transition metals can exhibit multiple oxidation states. In such cases, the oxidation state is indicated in Roman numerals in parentheses immediately following the metal name. For example, iron(III) chloride for FeCl3 indicates that iron has an oxidation state of +3.
Step-by-Step Guide to Naming Organic Compounds
Organic compound naming, especially for more complex molecules, requires a methodical approach:
Identify the Longest Carbon Chain
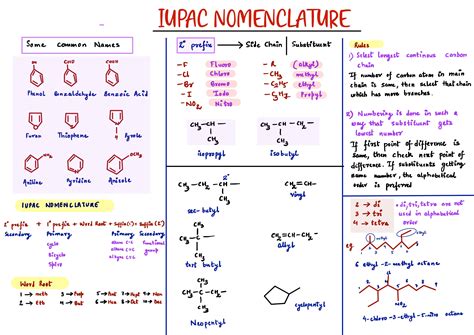
The first step in naming an organic compound is to identify the longest continuous chain of carbon atoms. This chain determines the base name of the compound, with the number of carbon atoms dictating the suffix.
Number the Carbon Chain
Number the carbon atoms in the chain starting from the end nearest to a substituent group. This helps in identifying the lowest set of locants (numbers indicating the positions of the substituents) for the substituents.
Identify and Name Substituents
Substituents attached to the carbon chain are named based on their parent group names with appropriate prefixes. For instance, a methyl group is named ‘methyl,’ and it is prefixed to the name of the base chain. Position numbers are added before the substituent name.
Combine Elements into One Name
Finally, the names of the substituents are listed in alphabetical order (excluding ‘mono’, ‘di’, ‘tri’ prefixes), followed by the base name of the chain, and the appropriate suffix indicating the functional group. For example, in 2-methylpropane, ‘2-methyl’ are the substituents, ‘propane’ is the base chain, and there are no additional suffixes needed.
Practical FAQ: Navigating Common Challenges
How do I decide on the base name for complex organic compounds?
For complex organic compounds, begin by identifying the longest continuous chain of carbon atoms that defines the base structure. The number of carbon atoms in this chain determines the suffix of the base name. For instance, if the longest chain has five carbon atoms, the base name will end in ‘-ane’, making it pentane. This foundational step helps in systematically naming the entire molecule.
What should I do when there are multiple functional groups in a compound?
When naming organic compounds with multiple functional groups, prioritize the primary functional group, which dictates the main class of the compound (like alcohol, aldehyde, ketone, etc.). Then, list the substituents in alphabetical order. For example, in 2,3-dibromo-1-chloropropane, ‘2,3-dibromo’ and ‘1-chloro’ are the substituents on the base chain ‘propane’.
Can I use common names instead of IUPAC names?
While common names are recognized in certain situations (especially in organic chemistry where traditional names are widely accepted), IUPAC names are preferred for their precision and clarity. Common names like ‘glycerol’ for propane-1,2,3-triol can be used in informal contexts or where confusion might arise with multiple isomers, but for scientific writing, IUPAC names are more appropriate.
Conclusion
Mastering IUPAC compound naming not only enhances your ability to communicate chemical structures clearly but also ensures that your scientific work adheres to international standards. By following the step-by-step guidance provided in this guide, you will be well-equipped to name a wide variety of chemical compounds accurately and confidently. Remember, practice makes perfect, so don’t hesitate to apply these principles to diverse compounds to deepen your understanding and proficiency.
This guide offers a comprehensive pathway from basic to advanced IUPAC naming principles. Whether you’re tackling simple or complex compounds, these actionable insights and practical tips will serve as your toolkit for precise and accurate scientific communication.