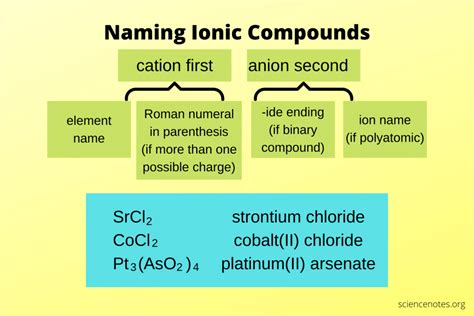
Naming ionic compounds is a fundamental skill in chemistry, critical for both academic success and practical applications in various fields such as pharmaceuticals and material sciences. This guide aims to elucidate the systematic approach to naming ionic compounds, providing clarity and practical insights for both students and professionals.
Understanding Ionic Compounds
Ionic compounds are formed when metals transfer electrons to nonmetals, creating positively charged cations and negatively charged anions that attract each other. Recognizing these components is the first step in naming an ionic compound. The formula typically reveals the ratio of cations to anions, which is pivotal in determining the name.Rules for Naming Ionic Compounds
When naming ionic compounds, it’s crucial to follow a specific set of rules. Initially, identify the cation and the anion. The name of the cation retains the element’s name, while the anion’s name ends in -ide. For example, in sodium chloride (NaCl), sodium (Na) is the cation, and chloride (Cl) is the anion.It's important to note that transition metals can have variable charges, so their names include the charge of the ion in Roman numerals. For example, iron (III) oxide (Fe2O3) names iron in the +3 oxidation state. Understanding these nuances is critical for accurate naming.
Key Insights
- Primary insight with practical relevance: Accurate naming of ionic compounds aids in precise communication and identification in both academic and industrial settings.
- Technical consideration with clear application: Knowledge of cation and anion names is essential for interpreting chemical formulas and understanding the properties and uses of the compounds.
- Actionable recommendation: Always double-check the charge states and names of cations and anions to ensure accurate and consistent naming.
Complex Ionic Compounds
Some ionic compounds contain polyatomic ions, which are groups of atoms that act as a single ion. These include common ions like sulfate (SO42-) and phosphate (PO43-). Understanding these complex ions can enhance comprehension and aid in accurate naming. For instance, in copper (II) sulfate pentahydrate (CuSO4·5H2O), the compound comprises a copper (II) ion, a sulfate ion, and five water molecules.The inclusion of hydrates, like pentahydrate in the example above, indicates the number of water molecules associated with each formula unit of the compound. This is important for both the identification and understanding of the compound’s physical properties.
Practical Examples
Consider magnesium fluoride (MgF2). Here, magnesium (Mg) is the cation with a +2 charge, and fluoride (F) is the anion with a -1 charge. The prefix “di-” indicates two fluoride ions. Another example is aluminum chloride (AlCl3). In this compound, aluminum (Al) is the cation with a +3 charge, and each chloride (Cl) ion carries a -1 charge.Let’s take calcium carbonate (CaCO3), an example frequently encountered in geological contexts. Here, calcium (Ca) is the cation, and carbonate (CO32-) is the anion. The name integrates directly from the ions’ names without any numerical prefix modifications.
Can transition metals have more than one ionic charge?
Yes, transition metals often exhibit variable charges depending on the elements they bond with. For instance, iron can form Fe2+ (ferrous) or Fe3+ (ferric) ions. Always include the charge in Roman numerals when naming transition metal compounds.
What if an ionic compound has more than one polyatomic ion?
When dealing with compounds containing multiple polyatomic ions, list each ion in the name following the naming conventions for each. For example, ammonium dihydrogen phosphate is (NH4)2 (H2PO4) indicating two ammonium ions and one dihydrogen phosphate ion.
By mastering these foundational principles and rules, chemists and students alike can navigate the intricacies of ionic compound nomenclature with confidence and precision.