Exploring the intricate world of chemistry reveals myriad compounds with unique properties and applications. One such fascinating compound is trifluoromethylamine, commonly known by its chemical name, nitrogen trifluoride (NF3). This seemingly simple molecule holds complex scientific significance. In this article, we delve into the intriguing aspects of NF3, examining its properties, applications, and relevance to modern chemistry.
Key Insights
- Nitrogen trifluoride is a colorless, non-flammable gas with significant applications in semiconductor manufacturing.
- Its unique molecular structure facilitates its use in plasma etching, an essential process in the fabrication of microelectronic devices.
- Understanding NF3’s environmental impact is crucial, as it is a potent greenhouse gas.
Chemical Properties of NF3
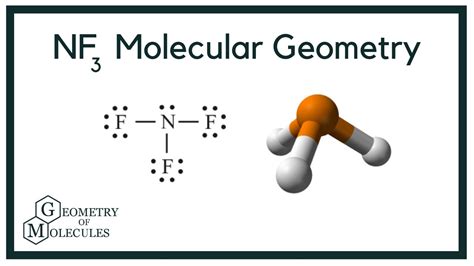
Nitrogen trifluoride (NF3) features a nitrogen atom bonded to three fluorine atoms in a molecular geometry that resembles a trigonal pyramid. The nitrogen atom’s lone pair contributes to the molecule’s polarity, which influences its reactivity and solubility in polar solvents. One of the standout features of NF3 is its high electronegativity, resulting in strong dipole-dipole interactions. These properties make NF3 a valuable reagent in chemical synthesis and industrial applications.Applications in Semiconductor Technology
NF3 has gained significant attention in the semiconductor industry. Its primary role comes from its use in plasma etching processes during the manufacturing of integrated circuits. Plasma etching involves the use of reactive gases to selectively remove materials from a substrate surface, achieving intricate patterns essential for modern microchips. NF3’s ability to form a self-limiting passivating oxide layer on silicon makes it a preferred choice for etching silicon-based materials, providing high selectivity and uniformity. Additionally, NF3 is utilized in cleaning processes for wafers, ensuring the removal of organic residues without damaging the substrate.The compound's low reactivity under normal conditions and its non-flammability further enhance its safety profile, allowing for its widespread adoption in cleanroom environments. Despite these benefits, NF3 presents some environmental concerns due to its high global warming potential. Understanding and mitigating its environmental impact remains a critical area of research for chemists and environmental scientists.
What is the environmental impact of NF3?
NF3 is a potent greenhouse gas with a global warming potential significantly higher than carbon dioxide. Its atmospheric release, primarily from semiconductor manufacturing, contributes to climate change. Efforts to reduce its emissions and explore alternatives with lower environmental impact are ongoing.
Why is NF3 used in plasma etching?
NF3 is used in plasma etching due to its excellent reactivity and selectivity towards silicon-based materials. Its self-limiting passivating oxide layer on silicon ensures uniform etching, which is crucial for high-quality microelectronic device fabrication.
To conclude, nitrogen trifluoride (NF3) stands as a pivotal compound in both chemical research and industrial applications, particularly in semiconductor manufacturing. Its unique chemical properties and role in plasma etching underscore its importance, while its environmental impact necessitates ongoing scrutiny and innovative solutions. As we advance in technology and environmental stewardship, understanding and optimizing NF3’s applications will remain vital.