Nitrogen monoxide, commonly referred to as nitric oxide (NO), plays a pivotal role in biological systems, industrial processes, and environmental science. Understanding its chemical formula, structure, and function can help unlock new pathways in both academic research and industrial applications. This article delves into the essentials of nitrogen monoxide, providing a thorough yet practical guide for professionals seeking to deepen their understanding.
Understanding the Chemical Formula and Structure
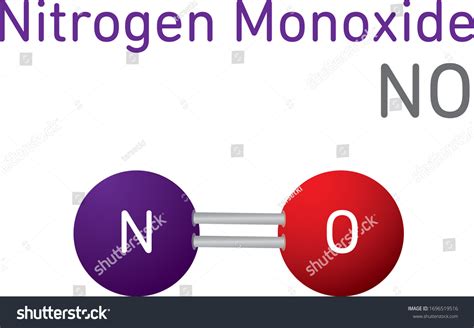
Nitrogen monoxide’s chemical formula is NO. It consists of one nitrogen (N) atom and one oxygen (O) atom. This molecular structure is simple yet crucial in comprehending its reactivity and role in various domains. NO is a diatomic molecule with a lone pair of electrons on the nitrogen atom, which accounts for its high reactivity. This free radical makes NO highly unstable and highly reactive, often participating in a variety of chemical reactions.
Applications in Biological and Industrial Fields
NO serves as a signaling molecule in the human body, playing a critical role in cardiovascular health by dilating blood vessels and preventing clot formation. In industrial settings, NO is integral in processes like nitric oxide therapy for pulmonary hypertension and in manufacturing, particularly in the control of pollutant emissions from combustion processes. For example, NO emissions are monitored and regulated in industrial environments to comply with environmental regulations and to reduce harmful effects on air quality.
The dual role of NO as both a beneficial biological agent and a pollutant exemplifies its complex nature. Understanding its behavior under different conditions is key to harnessing its potential benefits while mitigating adverse effects.
Key Insights
- Primary insight with practical relevance: NO’s dual role as both a beneficial biological agent and a pollutant requires precise management and understanding.
- Technical consideration with clear application: Monitoring NO emissions in industrial settings is crucial for compliance with environmental regulations.
- Actionable recommendation: Employ advanced monitoring technologies to manage NO levels effectively in both biological and industrial contexts.
How does nitric oxide function in the human body?
In the human body, nitric oxide (NO) acts as a signaling molecule that helps to regulate blood flow. It dilates blood vessels, improves blood flow, and plays a significant role in immune response mechanisms.
What are the environmental regulations for NO emissions?
Environmental regulations for NO emissions are governed by various national and international standards to control pollution from industrial processes. Agencies like the EPA (Environmental Protection Agency) in the U.S. set limits to ensure air quality and human health protection.
This exploration of nitrogen monoxide highlights its significant relevance across different fields, emphasizing its dual nature as both a beneficial molecule and a pollutant. By understanding its chemical characteristics and practical applications, professionals can better manage its effects and leverage its benefits in biological and industrial contexts.