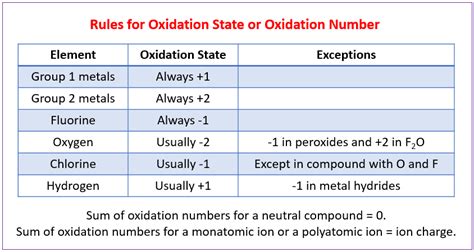
Understanding and mastering oxidation state rules is essential for anyone diving deep into the world of chemistry. Whether you’re an aspiring chemist, an educator, or a student, this guide will arm you with step-by-step guidance, actionable advice, and practical solutions to grasp these rules effectively. We’ll cover the basics and progress to more advanced concepts, addressing common challenges and providing solutions throughout.
Introduction to Oxidation States: Why It Matters
Oxidation states are a fundamental concept in chemistry that play a crucial role in predicting how chemical reactions will proceed. They allow us to understand the distribution of electrons in compounds and how atoms will bond with each other. Mastery of oxidation states will enhance your ability to predict reaction outcomes, balance chemical equations, and comprehend complex redox processes.
The Problem: Why Understanding Oxidation States is Challenging
Many students and professionals find it difficult to master oxidation states due to the seemingly abstract nature of the concept. The rules are nuanced, and applying them requires practice and a solid understanding of electron behavior. Moreover, tracking oxidation states during complex reactions can be daunting without a systematic approach.
In this guide, we aim to demystify the concept of oxidation states. We will break down the rules into simple, actionable steps and provide real-world examples to ensure you can apply these principles effectively.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by assigning oxidation states to elements in simple compounds to build a foundational understanding.
- Essential tip with step-by-step guidance: When assigning oxidation states, remember that hydrogen typically has an oxidation state of +1, and oxygen typically has an oxidation state of -2.
- Common mistake to avoid with solution: Avoid confusion between oxidation states and formal charges. Make sure to track changes in oxidation states during reactions for accurate results.
Detailed How-To: Assigning Oxidation States
Let’s delve into the mechanics of assigning oxidation states. The goal here is to systematically determine the oxidation state for each element in a compound.
The general steps include:
- Identify simple ions: Start with compounds containing simple ions such as sodium chloride (NaCl). Sodium (Na) has an oxidation state of +1, and chlorine (Cl) has an oxidation state of -1.
- Apply rules for common elements: Remember the standard oxidation states for common elements like hydrogen (+1), oxygen (-2), and halogens (usually -1). For example, in H2O, hydrogen is +1 and oxygen is -2.
- Calculate for polyatomic ions: For polyatomic ions such as sulfate (SO42-), start by recognizing the overall charge of the ion. Sulfur (S) typically has an oxidation state of +6 when bonded to oxygen atoms. The four oxygen atoms each have an oxidation state of -2, so the sum of these oxidation states is -8, making the sulfur oxidation state +6 to balance the -2 charge of the ion.
- Practice with complex compounds: Apply these rules to more complex compounds. For instance, in potassium permanganate (KMnO4), potassium (K) is +1, and the permanganate ion (MnO4-) has an overall charge of -1. Within the permanganate ion, manganese (Mn) has an oxidation state of +7.
Through practice, you'll become more adept at recognizing patterns and calculating oxidation states quickly.
Detailed How-To: Balancing Redox Reactions
Balancing redox (reduction-oxidation) reactions involves determining how oxidation states change and ensuring the number of electrons lost equals the number gained.
- Identify the oxidation states: Start by assigning oxidation states to all elements in the reactants and products. For example, in the reaction where zinc reacts with copper(II) sulfate to produce copper and zinc sulfate, determine the oxidation states of Zn, Cu, and S.
- Write the half-reactions: Separate the redox reaction into two half-reactions: one for the reduction and one for the oxidation. In this example, the half-reactions are:
- Reduction: Cu2+ (aq) + 2e- → Cu (s)
- Oxidation: Zn (s) → Zn2+ (aq) + 2e-
- Balance the atoms: Ensure both half-reactions have the same number of atoms of each element. Balance the non-oxygen, non-hydrogen atoms first.
- Balance the oxygen atoms: Use water (H2O) to balance oxygen atoms. In the above example, no additional balancing for oxygen is needed.
- Balance the hydrogen atoms: Use hydrogen ions (H+) to balance hydrogen atoms. Again, in this example, no balancing for hydrogen is required.
- Balance the charges: Balance the overall charge by adding electrons. The reduction half-reaction has a charge of +2, and the oxidation half-reaction has a charge of +2. To balance the charges, multiply the reduction half-reaction by 1 and the oxidation half-reaction by 1.
- Combine the half-reactions: Add the two balanced half-reactions together to obtain the balanced redox reaction.
The balanced reaction is:
Cu2+ (aq) + Zn (s) → Cu (s) + Zn2+ (aq)
Practical FAQ
Common user question about practical application
What should I do if I encounter complex polyatomic ions with multiple oxidation states?
When dealing with complex polyatomic ions, start by assigning an oxidation state to the group as a whole, based on its overall charge. Then, distribute the remaining charge among the individual atoms in the group based on standard oxidation states and the overall charge. For example, in dichromate (Cr2O72-), assume an overall charge of -2 for the ion. Chromium (Cr) typically has an oxidation state of +6. Since there are two chromium atoms, the total oxidation state for Cr is +12. There are seven oxygen atoms each with an oxidation state of -2, so the total for oxygen is -14. To balance this, the total charge of +12 from Cr and -14 from O results in the overall charge of -2 for the ion. This distribution of charge ensures the compound’s overall charge is balanced.
What is the best approach for remembering the common oxidation states of transition metals?
Transition metals often exhibit variable oxidation states, which can make them challenging to remember. One effective approach is to memorize the most common oxidation states for each element and practice identifying them in compounds. For example, iron (Fe) typically has oxidation states of +2 and +3, while copper (Cu) can be in +1 and +2 states. Create flashcards or use mnemonic devices to remember these common states and regularly practice assigning oxidation states to transition metal compounds.
How do I know when to use oxidation states versus formal charges?
Oxidation states and formal charges both describe electron distribution but serve different purposes. Use oxidation states to determine how electrons are distributed in compounds and track changes during redox reactions. Formal charges, on the other hand, help determine the best Lewis structure for a molecule by ensuring the sum of formal charges equals the actual charge of the molecule. To distinguish, remember that oxidation states track electron changes in the context of the entire compound, while formal charges are used for balancing