Demystifying the Complex Chemical Geometry of PO43 Lewis Structure
The exploration of PO43 Lewis structure, or the phosphate ion, offers a profound understanding of how chemical bonds and valence electrons are distributed within a molecule. This examination is essential for chemists as it provides insight into the molecular geometry and electronic configuration of phosphate, a critical compound in biochemistry, particularly in cellular energy transfer and nucleic acid structure. The focus here is to dissect the intricacies of the PO43 Lewis structure, its formation, and the resulting geometry, all presented through an expert lens, with practical insights and real examples.
Key Insights
- The PO43 Lewis structure reveals the electron distribution, aiding in understanding the phosphate ion's geometry.
- Understanding this structure highlights the importance of formal charges and resonance forms in depicting chemical reality.
- Knowledge of the PO43 Lewis structure is fundamental for predicting reactivity and interactions in biochemical processes.
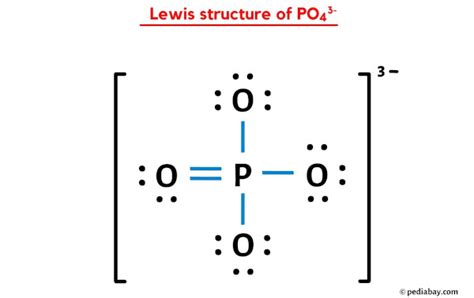
Phosphate ions (PO43-) possess a central phosphorus atom covalently bonded to four oxygen atoms. The process of drawing the Lewis structure begins by considering the total valence electrons—phosphorus contributes 5, and each of the four oxygens contributes 6. This totals to 32 valence electrons. To minimize electron-pair repulsion, phosphorus adopts an octet configuration with the help of the additional electrons from the three lone pairs required to make up the charge of -3.
The Lewis structure comprises one single, one double, and two triple-bonded oxygen atoms around phosphorus. Each double bond reduces the formal charge of the oxygen atom involved, while the phosphorus atom maintains a zero formal charge. To balance the -3 charge, three lone pairs are placed on the oxygen atoms, strategically ensuring the least repulsion and the most stable arrangement.
The PO43 Lewis structure is a pivotal aspect of comprehending the geometry of phosphate ions. Phosphate adopts a tetrahedral molecular geometry due to the arrangement of the four oxygen atoms around the central phosphorus. This geometry minimizes electron repulsion, adhering to VSEPR (Valence Shell Electron Pair Repulsion) theory. The bond angles within a perfect tetrahedral arrangement are 109.5 degrees, although slight deviations can occur due to the electronegativity differences between phosphorus and oxygen atoms.
In an analytical context, the PO43 Lewis structure plays a critical role in understanding phosphate’s behavior in various chemical reactions. For example, in biological systems, the phosphate group’s geometry and electron distribution affect ATP (adenosine triphosphate) formation and hydrolysis. The understanding of this structure is pivotal in fields such as medicinal chemistry, where phosphate analogs are designed for therapeutic interventions.
Why is the PO43- Lewis structure important in biochemistry?
The PO43- Lewis structure is crucial in biochemistry because it reveals how phosphate ions form and interact within biological systems. It helps explain the stability and reactivity of phosphate compounds, which are central to cellular energy storage and transfer, DNA and RNA structure, and various metabolic pathways.
Can the PO43 Lewis structure predict the phosphate ion's reactivity?
Yes, the PO43 Lewis structure provides insight into the phosphate ion's reactivity. By understanding the electron distribution and formal charges, one can predict how the phosphate ion will interact with other molecules, making it essential for drug design and understanding enzymatic reactions where phosphate groups play a role.
This analysis of the PO43 Lewis structure elucidates the fundamental principles behind the chemical geometry and reactivity of phosphate ions. Armed with this knowledge, one can better appreciate the intricate roles these ions play in both synthetic chemistry and biological systems. Through understanding the distribution of electrons and the resultant geometry, chemists can predict molecular behavior, facilitating advances in pharmaceuticals, agriculture, and beyond.