Understanding the root mean square speed (RMS speed) is crucial for anyone delving into kinetic theory of gases, thermodynamics, and statistical mechanics. RMS speed provides valuable insights into the velocity distribution of particles in a gas, helping us unravel the microscopic behavior underlying macroscopic observations. This article explores the concept in an accessible yet rigorous manner, providing practical insights for science enthusiasts.
Key Insights
- RMS speed is a key metric in kinetic theory of gases for understanding particle velocity distributions.
- Calculating RMS speed involves a square root of the average of the squared velocities of the gas particles.
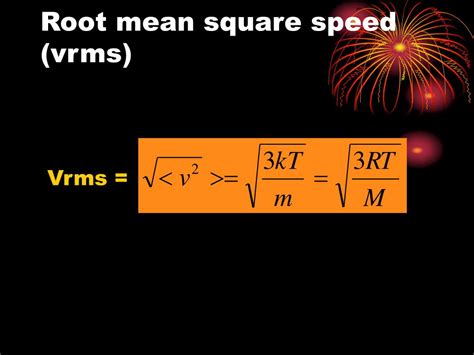
- To determine RMS speed, accurately measure the velocity distribution and apply the formula v_{rms} = \sqrt{\frac{3RT}{M}} , where R is the ideal gas constant, T is temperature, and M is molar mass.
The Foundation of RMS Speed
Root mean square speed is a statistical measure that describes the average speed of particles in a gas. Unlike mean speed, which averages the speeds directly, RMS speed accounts for the distribution of speeds by squaring each speed before averaging, thus giving more weight to higher speeds. This approach allows for a more accurate representation of the kinetic energy present within the gas.For a practical understanding, imagine a room filled with molecules of a gas. Each molecule moves at a different speed due to its kinetic energy. To capture this variability, RMS speed provides a unified measure that correlates directly with the thermal energy of the gas. In mathematical terms, RMS speed (( v{rms} )) is derived using the formula ( v{rms} = \sqrt{\frac{3kT}{m}} ), where ( k ) is Boltzmann’s constant and ( m ) is the mass of a single molecule. This formula underscores the dependency of RMS speed on temperature and mass.
Applications in Thermodynamic Studies
One of the most significant applications of RMS speed is in the study of gases within thermodynamics. Knowing the RMS speed enables scientists to predict gas behavior under different conditions. For example, RMS speed is crucial in determining how gases expand, mix, and react with other substances.Consider the ideal gas law, ( PV=nRT ), which governs the state of an ideal gas. Here, RMS speed helps in understanding how changes in temperature ( T ) and pressure ( P ) affect gas behavior. A higher temperature increases the average kinetic energy of gas particles, thus raising their RMS speed. This increase in kinetic energy directly influences reaction rates, diffusion, and even the volume occupied by the gas.
Moreover, in practical applications such as engine design and environmental modeling, RMS speed calculations aid in refining predictions about how pollutants disperse in the atmosphere or how heat is transferred through gaseous systems.
What is the significance of RMS speed in real-world applications?
RMS speed is vital in various fields such as engineering, environmental science, and industrial chemistry. It helps predict gas behavior in engines, assess pollutant dispersion, and design better thermal insulation.
How does changing the temperature affect RMS speed?
An increase in temperature leads to an increase in RMS speed, as it raises the kinetic energy and thus the velocities of gas particles. Conversely, lowering the temperature will decrease the RMS speed.
In conclusion, RMS speed serves as an indispensable tool in both theoretical and practical scientific endeavors. Its role in kinetic theory, thermodynamics, and practical applications underscores its importance for science enthusiasts and professionals alike. Through an in-depth understanding and accurate calculations, RMS speed unveils the microscopic dynamics governing macroscopic observations.