Understanding the SF4 Lewis Structure unveils critical insights into molecular geometry and chemical bonding. This sulfur tetrafluoride molecule’s electron configuration offers practical knowledge that extends beyond theoretical chemistry into various applications like materials science and drug design.
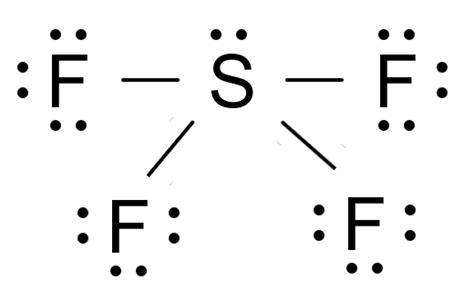
To grasp the intricacies of SF4’s structure, one must dive deep into valence electrons, lone pairs, and formal charge calculations. SF4 consists of a central sulfur atom surrounded by four fluorine atoms, presenting a unique case in the realm of molecular geometries.
Key Insights
- Primary insight with practical relevance: The SF4 Lewis structure reveals the importance of lone pairs in determining the molecule's unique see-saw shape.
- Technical consideration with clear application: The arrangement of electrons affects the molecule’s polarity and its interactions in chemical reactions.
- Actionable recommendation: Use the VSEPR theory to predict the molecular shape accurately.
Understanding the SF4 Lewis structure necessitates a deep dive into its valence electrons. Sulfur, belonging to group 16, has six valence electrons. Fluorine, from group 17, each has seven valence electrons. In SF4, sulfur forms four covalent bonds with four fluorine atoms, leaving two valence electrons to be used for lone pairs. According to the Lewis structure, sulfur central atom has a total of ten electrons (forming four single bonds with four fluorine atoms and having two lone pairs). These lone pairs are pivotal in dictating the molecule’s geometry.
Electron Configuration and Lone Pairs
SF4’s electron configuration is crucial for comprehending its molecular shape. The presence of lone pairs on the central atom influences the bond angle and overall molecular geometry. In SF4, the lone pairs occupy equatorial positions in a trigonal bipyramidal arrangement, minimizing repulsion while maintaining stability. These lone pairs create a see-saw shape, characterized by a 90-degree angle between the lone pair and axial bonds, which contrasts with the 180-degree angle between the axial bonds. This geometry provides insight into how lone pairs on central atoms affect molecular geometry.Application in Chemical Reactions
The SF4 Lewis structure’s geometry and electronic configuration have significant implications for its chemical reactivity. The see-saw geometry implies an asymmetric charge distribution, contributing to the molecule’s polarity. Polar molecules like SF4 can engage in dipole-dipole interactions and hydrogen bonding (though less common here), influencing the compound’s solubility in polar solvents. This property is beneficial in designing materials where selective chemical reactivity is desired, such as catalysts in fine chemical synthesis.Why is the SF4 molecule's shape described as a see-saw?
The molecule adopts a see-saw shape due to the placement of two lone pairs on the equatorial plane of a trigonal bipyramidal arrangement, which minimizes repulsion between the lone pairs and the bonding pairs.
How does the polarity of SF4 affect its chemical behavior?
The polarity of SF4, due to its asymmetrical structure, influences its interaction with other polar molecules, enhancing its solubility in polar solvents and its reactivity in chemical reactions.
Understanding the SF4 Lewis structure not only bridges theoretical knowledge and practical applications but also serves as a model for more complex molecules. Mastery of such structures is indispensable for chemists, material scientists, and researchers working on the forefront of molecular chemistry.