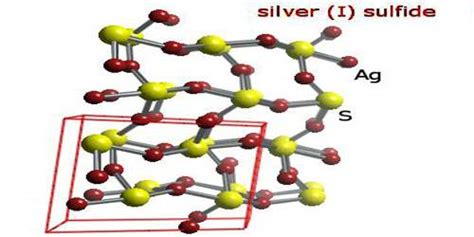
Understanding the complexities of the silver sulfide formula requires delving into both its chemical composition and practical applications. This compound, with the formula Ag2S, is a significant component in various industries including electronics and photography. To demystify this formula and unveil its intricate nuances, it is crucial to explore its properties, behavior in different environments, and real-world uses. This article aims to illuminate these aspects, providing a robust understanding for professionals and enthusiasts alike.
Key insights box:
Key Insights
- Ag2S exhibits exceptional properties making it invaluable in semiconductor applications.
- The formula’s sensitivity to light is a fundamental aspect impacting its use in photosensitive materials.
- Strategic synthesis methods enhance its usability in modern technological advancements.
Ag2S, also known as silver sulfide, has captured interest due to its unique optical and electronic properties. The compound’s ability to function as a semiconductor makes it a critical material in the production of photovoltaic cells. Its application in converting sunlight into electrical energy highlights the practical relevance of its semiconductor capabilities. In semiconductor technology, Ag2S’s role is amplified by its excellent conductivity, making it indispensable for various electronic devices.
The behavior of Ag2S under different environmental conditions is another crucial aspect. The compound is highly sensitive to light, which presents both challenges and opportunities. This light sensitivity is exploited in photographic materials where controlled exposure to light can result in precise image formation. The interaction between Ag2S and light is pivotal in these applications, demonstrating its practical utility. In contrast, its reactivity under oxidative conditions must be meticulously managed to avoid degradation, which can affect its performance in long-term applications.
Synthesis of silver sulfide presents several technical considerations. One of the most effective methods is the reaction between silver nitrate (AgNO3) and sodium sulfide (Na2S) in an aqueous solution. The method results in the formation of Ag2S precipitates. Understanding the controlled parameters, such as temperature and pH, is essential to obtain high-purity Ag2S crystals, which are crucial for advanced applications. The synthesis process also underscores the importance of precise instrumentation and conditions to ensure the compound’s optimal functionality.
FAQ section:
What are the primary applications of silver sulfide?
Ag2S finds extensive use in semiconductor technology, especially in photovoltaic cells due to its semiconductor properties. It is also utilized in photographic materials because of its light-sensitive nature, which allows for precise image formation.
How should the stability of silver sulfide be managed in different environments?
The stability of Ag2S needs careful management, especially when exposed to light and oxidative conditions. Maintaining the compound in a controlled environment, shielding from direct light, and using protective coatings can help mitigate its degradation, thereby preserving its integrity for long-term applications.
Ag2S holds substantial promise across diverse fields, thanks to its unique properties and practical applications. By understanding its chemical behavior and leveraging its potential, professionals can unlock new possibilities in semiconductor technology, electronic devices, and beyond. This exploration underscores the importance of a thorough grasp of silver sulfide’s formula and its multifaceted applications.