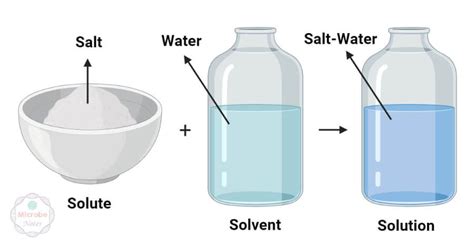
Understanding solute definition biology is essential for grasping the fundamentals of chemistry and biology. A solute is a substance that is dissolved in another substance, known as a solvent, creating a solution. This concept forms the bedrock of many biological processes, from cellular transport to metabolic functions. To fully appreciate the importance and practical applications, let’s delve deeper into what solutes are, why they matter, and how they operate within biological systems.
Key Insights
- A solute is a substance dissolved in a solvent to form a solution, crucial for many biological processes.
- Electrolytes, which are types of solutes that ionize in solution, play a vital role in maintaining cellular function.
- Understanding solute interactions can lead to effective treatments in medicine and healthcare.
Understanding Solutes: Basic Chemistry
At its core, a solute is a substance that, when mixed with a solvent, dissolves to form a homogeneous mixture or solution. This fundamental concept applies universally, whether we are talking about salt in water or glucose in blood. The properties of solutes dictate the behavior of solutions, impacting viscosity, temperature, and even the phase changes of the mixture. In biological contexts, solutes interact with solvents in ways that facilitate complex processes like nutrient uptake, waste elimination, and homeostasis.
The Role of Solutes in Biological Systems
Solute definition biology becomes especially pertinent when considering the human body. Here, solutes are primarily composed of water, electrolytes, and dissolved gases. These substances interact to maintain a balance necessary for physiological functions. For example, electrolytes like sodium, potassium, and calcium are critical for nerve transmission and muscle contraction. Disruption in their balance can lead to severe health issues, underscoring the importance of solute definition biology in medical science. An understanding of solute dynamics enables scientists to develop targeted therapies, such as those for conditions involving electrolyte imbalances or renal dysfunction.
What are the common examples of solutes in the human body?
Common examples include electrolytes like sodium, potassium, and calcium, which play crucial roles in nerve function, muscle contraction, and maintaining fluid balance.
How do solutes affect the osmotic balance in cells?
Solute concentration impacts osmotic pressure, which drives water movement across cell membranes. Maintaining the right solute concentrations is essential for cellular functions and overall health.
In conclusion, a deep understanding of solute definition biology is invaluable, whether for advanced scientific research or everyday health considerations. Knowing how solutes interact within solvents allows us to better understand and treat biological systems. Whether you are a medical professional, a biologist, or simply curious, grasping these principles can illuminate the intricate workings of life itself.