Understanding the volume formula in chemistry is crucial for anyone working in the field, whether you’re a student, an aspiring chemist, or someone involved in laboratory research. Mastering this concept will not only help you excel in your chemistry coursework but also equip you with practical skills that are invaluable in real-world applications. This guide will walk you through the intricacies of the volume formula, addressing common pain points and providing actionable advice for a deeper understanding.
The Importance of the Volume Formula in Chemistry
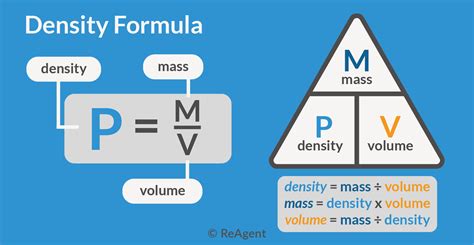
The volume formula is fundamental in chemistry, serving as the backbone for calculations involving gases, liquids, and solids. Understanding this formula allows you to determine the amount of space a substance occupies, which is essential for stoichiometric calculations, reaction proportions, and various other chemical processes. Whether you’re working on a classroom project or a research study, a strong grasp of volume concepts can streamline your experiments and enhance your analytical capabilities.
Many beginners struggle with the volume formula because it combines elements of algebra and basic chemistry principles. However, by breaking down this formula into manageable steps and applying real-world examples, you can conquer these challenges.
Quick Reference Guide
Quick Reference
- Immediate action item with clear benefit: Calculate the volume of a gas using the ideal gas law formula, PV=nRT. This can help you determine the volume under given conditions and is crucial for reaction monitoring.
- Essential tip with step-by-step guidance: When calculating the volume of a liquid, always use a graduated cylinder and measure accurately. Ensure to convert all units to SI units for consistency.
- Common mistake to avoid with solution: Mixing up units is a frequent error. Double-check all conversions to ensure the final volume is in the correct units (e.g., liters, milliliters).
Mastering Gas Volume Calculations
Gas volume calculations are often daunting, but with systematic approach and practical examples, they can become straightforward.
To calculate the volume of a gas, we often use the ideal gas law:
PV=nRT
Where:
- P is the pressure in atmospheres (atm)
- V is the volume in liters (L)
- n is the number of moles of the gas
- R is the universal gas constant (0.0821 L·atm/mol·K)
- T is the temperature in Kelvin (K)
Let’s walk through an example to illustrate this formula in action:
Imagine you have a gas sample with the following conditions:
- Pressure (P) = 2.5 atm
- Number of moles (n) = 0.3 mol
- Temperature (T) = 300 K
You need to find the volume (V). Plug the values into the ideal gas law equation:
V = nRT/P
V = (0.3 mol) * (0.0821 L·atm/mol·K) * (300 K) / (2.5 atm)
V = 2.96 L
Thus, the volume of the gas sample is 2.96 liters under the given conditions.
This step-by-step breakdown demonstrates the practical application of the ideal gas law, allowing you to confidently calculate gas volumes for any set of given conditions.
Calculating Liquid and Solid Volumes
Volume calculations for liquids and solids often involve straightforward geometric principles. Let’s explore the basic formulas for common shapes and apply them with practical examples.
For liquids, one of the simplest methods to measure volume is using a graduated cylinder:
Procedure:
- Place the graduated cylinder on a flat surface.
- Pour the liquid slowly into the cylinder, minimizing splashing.
- View the meniscus at eye level to read the volume accurately.
Suppose you need to measure the volume of a liquid. Here’s how:
If you read 15.7 mL at the bottom of the meniscus, that’s the volume of your liquid.
For solids, geometric formulas come into play. Here are some common shapes:
- Cube: Volume (V) = side length^3
- Rectangular Prism: Volume (V) = length * width * height
- Sphere: Volume (V) = 4/3 * π * radius^3
Let’s consider a few practical examples:
Example 1: Cube
If each side of a cube measures 3 cm:
V = 3 cm * 3 cm * 3 cm
V = 27 cubic centimeters (cm³)
Example 2: Rectangular Prism
For a rectangular block with dimensions 5 cm (length) by 4 cm (width) by 2 cm (height):
V = 5 cm * 4 cm * 2 cm
V = 40 cubic centimeters (cm³)
Example 3: Sphere
If a sphere has a radius of 2 cm:
V = 4/3 * π * (2 cm)^3
V ≈ 33.51 cm³
Practical FAQ Section
How do I convert volume units accurately?
To convert volume units, you should be familiar with basic conversion factors. Here are a few common conversions:
- 1 liter (L) = 1000 milliliters (mL)
- 1 cubic meter (m³) = 1,000,000 liters (L)
- 1 gallon (gal) = 3.785 liters (L)
For example, if you have 500 mL and need to convert it to liters:
500 mL ÷ 1000 = 0.5 L
Remember, always double-check your calculations to ensure accuracy.
Why is it important to maintain accuracy when measuring volumes?
Accuracy in volume measurements is critical for several reasons:
- Stoichiometric calculations: Precise volumes ensure accurate determination of reactants and products.
- Concentration determination: Accurate volumes allow for precise preparation of solutions.
- Experiment reproducibility: Consistent measurements are vital for reproducing experimental results.
Inaccurate measurements can lead to incorrect results, wasted resources, and complications in further experiments.
Can you provide an example of using volume to calculate reaction yield?
Certainly! Let’s consider a reaction where gas volumes are involved:
Suppose you have a reaction between hydrogen and oxygen to form water:
2H₂(g) + O₂(g) → 2H₂O(g)
You have 4 L of hydrogen gas and 2 L of oxygen gas at standard temperature and pressure (STP). To determine the limiting reactant and calculate the volume of water produced, follow these steps:
- Identify the stoichiometric ratios:
- 2 moles of H₂