Understanding the concept of main group elements is critical for anyone delving into the study of chemistry. This guide will demystify what main group elements are, their characteristics, and how they play a pivotal role in various chemical reactions and properties. Whether you’re a student, a professional, or just someone with a keen interest in chemistry, this guide will serve as your comprehensive resource to grasp these fundamental components of the periodic table.
What Are Main Group Elements?
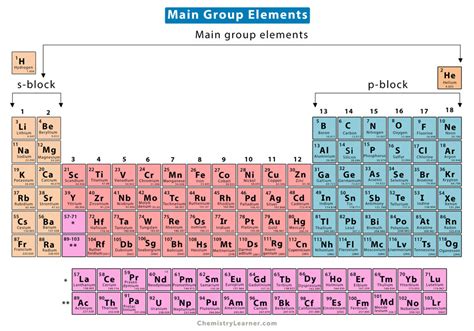
Main group elements, also referred to as representative elements, are the elements located in groups 1, 2, and 13 through 18 of the periodic table. They include the alkali metals, alkaline earth metals, and the p-block elements. These elements are significant because they are the most reactive and often form the basis for numerous chemical compounds and reactions.
Why Should You Care About Main Group Elements?
Understanding main group elements is crucial for several reasons:
- They form the basis of many everyday substances, from the water we drink to the materials we use in construction.
- They participate in a wide array of chemical reactions, making them central to both organic and inorganic chemistry.
- Knowledge of these elements can help in the fields of materials science, environmental science, and industrial chemistry.
If you are looking to solve practical chemistry problems or understand the chemical composition of materials around you, having a solid grasp of main group elements is essential.
Quick Reference
Quick Reference
- Immediate action item: Start with memorizing the location of main group elements on the periodic table.
- Essential tip: Learn the valence electron configuration for each main group element.
- Common mistake to avoid: Confusing main group elements with transition metals; ensure you recognize the different reactivity patterns.
Detailed Guide: Understanding Main Group Elements
Main group elements are characterized by their full s and p orbitals. This electron configuration is responsible for their high reactivity and ability to form a variety of compounds. Here’s a deep dive into what distinguishes these elements:
The Structure of Main Group Elements
The main group elements are categorized by their group numbers:
- Group 1: Alkali Metals – These elements, including lithium (Li), sodium (Na), and potassium (K), are highly reactive, especially with water. They always have one electron in their outermost shell.
- Group 2: Alkaline Earth Metals – Elements like beryllium (Be), magnesium (Mg), and calcium (Ca) are less reactive than alkali metals but still react vigorously with water. They have two electrons in their outermost shell.
- Group 13 to 18: P-Block Elements – These include elements like boron (B), carbon ©, nitrogen (N), oxygen (O), and others up to helium (He). Their reactivity varies greatly, from highly reactive to inert, depending on their group.
Reactivity and Compound Formation
Main group elements easily form ions due to their electron configurations. Alkali and alkaline earth metals lose electrons to form positive ions, while p-block elements either gain or share electrons to form negative ions or covalent compounds.
For example, sodium (Na) from Group 1 loses one electron to form a sodium ion (Na+), while chlorine (Cl) from Group 17 gains one electron to form a chloride ion (Cl-). These ions can then combine to form compounds such as sodium chloride (NaCl), a fundamental ingredient in our everyday diet.
Valence Electron Configuration
Understanding the valence electron configuration is crucial for predicting the chemical behavior of main group elements:
| Group | Outermost Shell Configuration |
|---|---|
| 1 (Alkali Metals) | ns1 |
| 2 (Alkaline Earth Metals) | ns2 |
| 13 (Boron Group) | ns2np1 |
| 14 (Carbon Group) | ns2np2 |
| 15 (Nitrogen Group) | ns2np3 |
| 16 (Oxygen Group) | ns2np4 |
| 17 (Halogens) | ns2np5 |
| 18 (Noble Gases) | ns2np6 |
Practical Tips for Handling Main Group Elements
Here are some practical tips to remember when dealing with main group elements:
- Always store alkali metals in oil to prevent them from reacting violently with water.
- Use proper PPE (Personal Protective Equipment) when handling reactive elements such as alkali and alkaline earth metals.
- Understand the stability and reactivity trends within the groups to predict compound formation and reaction outcomes.
FAQ: Common User Questions About Main Group Elements
Why Are Main Group Elements So Reactive?
Main group elements are highly reactive because they have incomplete outer electron shells. To achieve a stable electron configuration similar to the noble gases, they readily gain, lose, or share electrons. For example, carbon in Group 14 can form four covalent bonds to fill its outer shell, making it a fundamental building block for organic molecules.
Can Main Group Elements Form Multiple Compounds?
Absolutely! Main group elements, especially those in the p-block, exhibit a remarkable ability to form diverse compounds. For example, carbon can form millions of different organic compounds through covalent bonding with other elements like hydrogen, oxygen, and nitrogen. This versatility is what allows carbon to serve as the backbone of life.
How Do I Identify a Main Group Element?
Identifying main group elements is straightforward if you’re familiar with the periodic table. They are located in groups 1, 2, and 13 through 18. To pinpoint a main group element, locate it on the periodic table, check its group number, and ensure it’s not a transition metal or an inner transition metal. Remember that main group elements are typically more reactive than their d-block counterparts.
By understanding the role and characteristics of main group elements, you are better equipped to tackle various chemical concepts and applications. From predicting reaction outcomes to identifying the elements in your everyday life, this guide should serve as a reliable resource for anyone curious about chemistry.