Understanding the fundamental differences between ionic and covalent bonds is critical for comprehending chemical structures and reactions. These bond types play a crucial role in the stability, properties, and behavior of substances at a molecular level. This article will delve into the intricacies of these bonds, offering practical insights, real-life examples, and evidence-based statements.
Key Insights
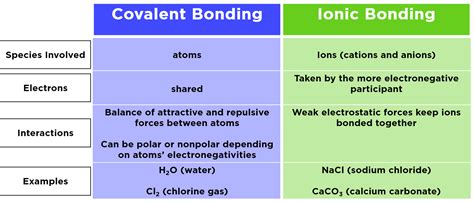
- Ionic bonds are formed through the transfer of electrons, leading to electrostatic attraction between oppositely charged ions.
- Covalent bonds occur when atoms share electrons, creating strong molecular bonds.
- Understanding these differences is essential for predicting molecular behavior, stability, and reactions.
Ionic bonds primarily form between metals and non-metals. When an atom donates an electron to another atom, it creates a positively charged ion (cation) and a negatively charged ion (anion). These ions are held together by strong electrostatic forces. This bond type is common in compounds such as sodium chloride (NaCl), where sodium (Na) donates an electron to chlorine (Cl). The resulting ionic lattice structure explains the high melting and boiling points of ionic compounds. Ionic bonds are also responsible for the solubility of these compounds in water due to the interaction between water molecules and the ions.
Covalent bonds, on the other hand, typically occur between non-metal atoms. In this bond type, atoms share electrons to fill their outer electron shells, achieving a more stable configuration. A classic example is water (H2O), where two hydrogen atoms share electrons with one oxygen atom. This sharing results in a molecule with a defined shape and polarity, influencing properties such as reactivity and phase states. The stability of covalent bonds allows compounds like glucose (C6H12O6) to maintain their structure in various environments.
Ionic Bonds: Formation and Characteristics
Ionic bonds are formed through the transfer of electrons from one atom to another, leading to the formation of charged particles known as ions. The driving force behind this bond is the electrostatic attraction between the positively charged cation and the negatively charged anion. For instance, in the formation of magnesium chloride (MgCl2), magnesium (Mg) loses two electrons to become Mg2+, while each chlorine (Cl) atom gains one electron to become Cl-. The resulting ionic compound is held together by the strong attraction between Mg2+ and 2Cl-. This attraction gives ionic compounds their high melting and boiling points and their characteristic solubility in polar solvents like water.
Covalent Bonds: Formation and Characteristics
Covalent bonds are formed by the sharing of electrons between atoms, allowing them to achieve a stable electron configuration. This type of bonding commonly occurs between atoms of non-metals. A prime example is the molecule of carbon dioxide (CO2), where one carbon atom shares electrons with two oxygen atoms. This sharing allows each atom to achieve a full outer shell of electrons, resulting in a molecule that is both stable and non-polar. The covalent nature of these bonds contributes to the properties of the substances involved; for example, the strength and rigidity of diamond, where each carbon atom shares electrons with four other carbon atoms in a tetrahedral arrangement, exemplify the strong covalent bonds within.
What is an example of an ionic compound?
A common example of an ionic compound is sodium chloride (NaCl), where sodium donates an electron to chlorine to form ions that attract each other.
How do covalent bonds differ from ionic bonds in terms of molecular structure?
In covalent bonds, atoms share electrons, creating stable molecules like water (H2O), whereas ionic bonds involve the transfer of electrons and the attraction between ions, forming structures like sodium chloride (NaCl).