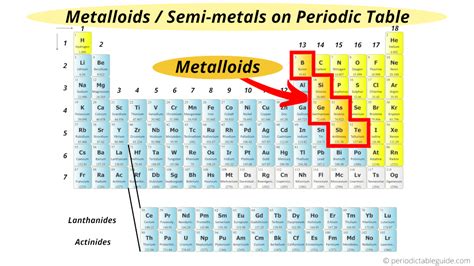
Understanding metalloids on the periodic table is crucial for grasping the nuanced differences in chemical behavior between metals and nonmetals. Metalloids, also known as semimetals, bridge the gap between metals and nonmetals, exhibiting properties of both categories. This makes them particularly fascinating from both theoretical and practical perspectives.
Key insights
Key Insights
- Metalloids display a blend of metallic and nonmetallic characteristics.
- They are situated in a diagonal region between metals and nonmetals on the periodic table.
- An actionable recommendation: Conduct specific research on boron, silicon, and arsenic for detailed practical applications.
Understanding the periodic table layout is essential for pinpointing metalloids accurately. Metalloids occupy a unique position in the periodic table, lying in a zigzag line between metals and nonmetals. This diagonal region, often referred to as the “staircase,” includes boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), and tellurium (Te). Each of these elements has unique properties that make them particularly valuable in various industrial and technological applications.
The diagonal location of metalloids signifies their unique ability to form alloys and compounds that blend the characteristics of both metals and nonmetals, contributing significantly to advanced material science. Silicon, for example, is a cornerstone in the semiconductor industry, while boron’s unique nuclear properties are vital in nuclear reactors and astrophysics.
Another practical insight lies in understanding how metalloids can be used in both electronic and biological fields. Arsenic, despite its toxicity, has been historically used in medicine and is currently being explored in nanomedicine. Germanium, a close relative to silicon, has seen resurgence in infrared optics.
In-depth analysis of metalloids’ positioning and properties
To delve deeper, the metalloids’ position on the periodic table is not just a random arrangement but a reflection of their unique electron configurations and bonding capabilities. They possess relatively high melting points and specific electrical conductivity, allowing them to behave in ways that neither fully metal nor fully nonmetal elements would. For instance, silicon’s ability to conduct electricity under certain conditions makes it indispensable in electronics. This duality makes metalloids invaluable in developing new technologies and improving existing ones.
Understanding their unique behavior is key to leveraging their properties in various fields. The ability of metalloids to act as semiconductors, for example, forms the foundation of modern electronic devices. The p-n junction in diodes and transistors, essential components in almost all electronic circuits, relies on the semiconducting properties of elements like silicon and germanium. This practical application underscores the importance of comprehending metalloids’ role within the periodic table.
Another essential aspect is their occurrence in nature and extraction. Metalloids are rarely found in their free form in nature due to their reactive nature. Instead, they are often extracted from various ores and minerals. The extraction process involves complex chemical procedures that highlight their reactive nature further. For instance, refining silicon from its ore involves reducing silicon dioxide (SiO2) with carbon in a high-temperature process.
FAQ section
What are the primary uses of metalloids in technology?
Metalloids such as silicon and germanium play critical roles in technology, especially in the semiconductor industry. Silicon is the primary material used in the manufacture of computer chips and solar cells. Germanium, due to its transparency to infrared light, is used in infrared optical systems.
Why are metalloids considered unique compared to metals and nonmetals?
Metalloids are unique due to their hybrid properties, which combine characteristics of both metals and nonmetals. They have a higher melting point than most nonmetals and display electrical conductivity that can vary between metallic and nonmetallic, making them crucial in technological advancements, especially in semiconductors.
In conclusion, metalloids’ distinctive properties and their strategic placement on the periodic table make them pivotal to both scientific understanding and technological innovation. Their unique blend of characteristics, combined with their practical applications, underscores their importance in advancing our technological capabilities.